EMBOLIZE: Bringing Randomized Evidence to Pelvic Vein Embolization
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of Cath Lab Digest or HMP Global, their employees, and affiliates.
CLD talks with Ronald S. Winokur, MD, FSIR, FAVLS, RPVI
Co-Principal Investigator, EMBOLIZE Study
Professor of Clinical Radiology; Director, Weill Cornell Vein Center, Weill Cornell Medicine, Division of Interventional Radiology, Department of Radiology, New York, New York
Can you tell us about pelvic venous disease?
Pelvic congestion, or pelvic venous disease (PeVD), is a chronic condition that causes pelvic pain in women. It tends to develop over time and often worsens with pregnancy. Many conditions can cause pelvic pain and symptoms can vary. As a result, women with PeVD are often misdiagnosed and may receive improper treatment.
What’s happening is that enlarged veins develop in the pelvis around the uterus or ovaries, which can lead to chronic pelvic pain. A lot of it relates to venous reflux, leading to “backup” in the ovarian and pelvic veins, very similar to varicose veins in the legs. The frustrating part has been that we’ve had good treatment options with pelvic vein embolization, but it has never really had a randomized controlled trial behind it, so physicians have been hesitant in adoption.
What challenges do patients face with getting diagnosed and treated?
There actually is a fair amount of awareness of the disease. The problem is that these patients still tend to get pushed aside. They’ll go to their OB/GYN, and the OB/GYN may do a hysterectomy or try something else, and the pain doesn’t improve.
Then they end up in this chronic “pelvic pain” world, doing physical therapy and other treatments that don’t really get them anywhere, without anyone asking whether there is an underlying venous etiology that should be evaluated and treated.
Can you tell us about the EMBOLIZE trial?
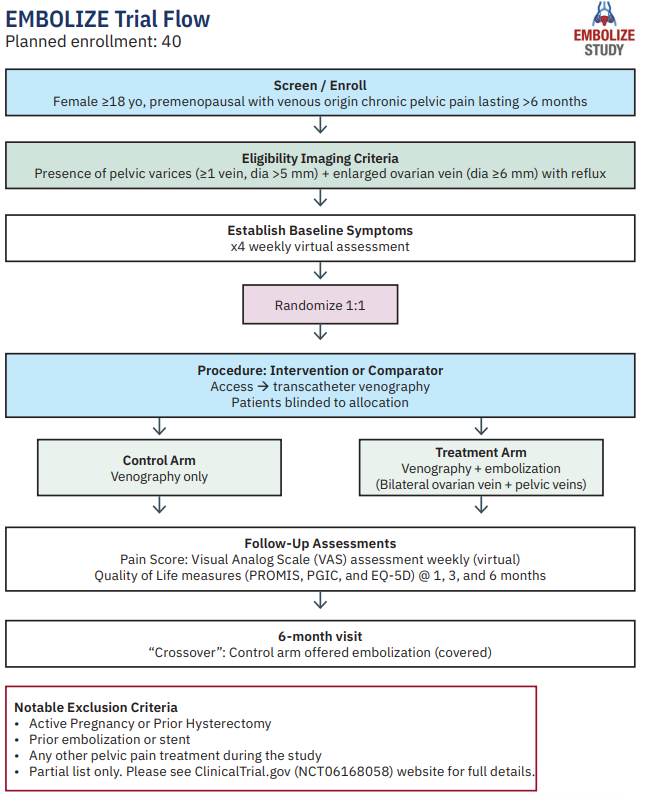
EMBOLIZE is a prospective, randomized, controlled trial studying the effects of ovarian vein and pelvic vein embolization in reducing pelvic pain in women due to PeVD (see flowchart). Patients are randomized to either venography alone or venography plus embolization.
In terms of eligibility, the imaging criteria include at least one dilated, incompetent ovarian vein (≥6 mm) and pelvic varices with at least one vein >5 mm. The intervention arm is venography plus embolization of the ovarian veins and pelvic veins/varices.
After the procedure, patients complete self-assessments weekly for six months, and then we also see them at 1, 3, and 6 months. Our primary endpoint is change in pain on a visual analog scale (VAS), and we’re also capturing quality of life and patient-reported improvement using validated instruments.
One important part of the design is that at six months, the subjects who were in the control arm can come back, undergo embolization, and have it covered by the study. Right now, we track all subjects for at least the first one to three months after the end of the study, regardless of which arm they were in, so we can combine outcomes for embolization.
Planned enrollment is 40 patients with study completion estimated for the end of 2027.
Why is this trial so important?
In other venous conditions, we have evidence. Procedures like venous stenting or laser closure of the great saphenous vein for varicose veins show good outcomes. In contrast, for PeVD, there’s very little data around pelvic vein embolization. With EMBOLIZE, we now are finally in the realm of proving efficacy, where we can show that pelvic vein embolization can be successful.
Because of a lack of consensus on diagnosis and treatment criteria, evidence-based treatments for this chronic pain are often excluded from health insurance policies in the United States. As a result, PeVD treatments are not always covered for women. This has led the condition to be under-recognized and untreated for countless women.
If we can show clear efficacy and safety in a rigorous trial, it gives payers something concrete to hang their hat on, and it can move this procedure toward more consistent, evidence-based coverage.
Can you walk us through your approach to pelvic vein embolization?
We plan ahead with imaging. As part of the EMBOLIZE trial, we are doing computed tomography (CT) scans performed using a CT venography (CTV) protocol and comparing that to ultrasound.
From a procedural standpoint, the left ovarian vein is often dilated and stretched so the valves can’t close. From there, we can go down the inferior vena cava into the left renal vein, and then down the left ovarian vein into the pelvis. We inject sclerosis medication into the dilated pelvic veins that are driving symptoms, and we place coils in the ovarian vein.
Then we come back and treat the right side with coils as well, so we are embolizing and addressing the whole pelvic venous network bilaterally.
As part of EMBOLIZE, we also evaluate the internal iliac veins, which are another major drainage pathway of the pelvis. We inject contrast, and if there are residual veins visible, and they weren’t already pre-closed with the initial injection, then those can be treated via the internal iliac system. Otherwise, we may inject additional sclerosis into the internal iliacs, but we don’t place coils there because we worry about coil migration.
Coil migration is one of the challenges of embolization. Historically, rates have been around 1% to 3%, and I think those events were largely in the internal iliacs and other locations that we are not including in this study.
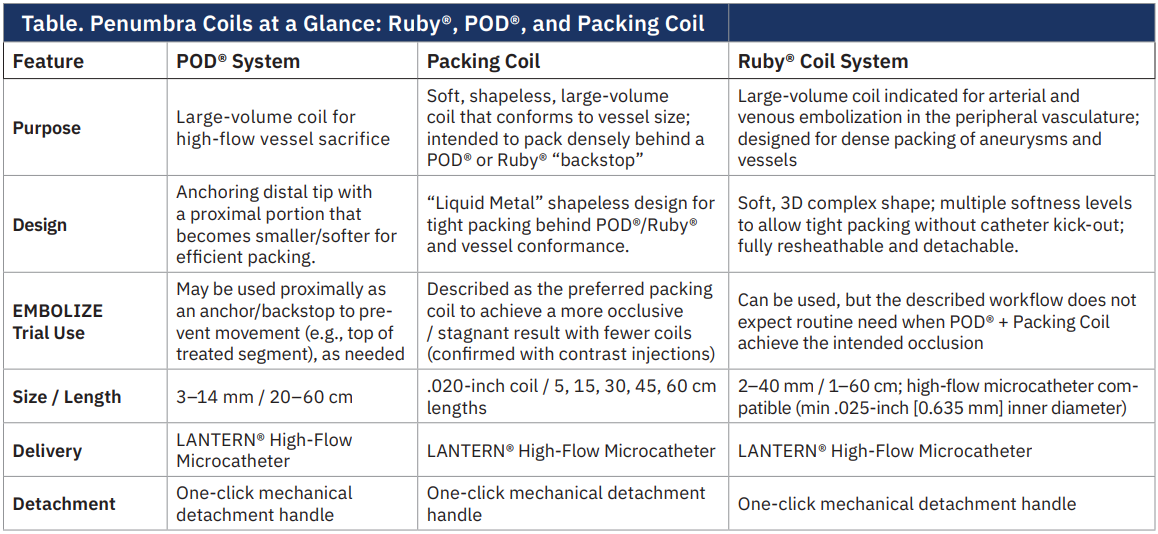
What are the benefits of Penumbra’s Ruby, POD, and packing coils that are being evaluated in the EMBOLIZE trial?
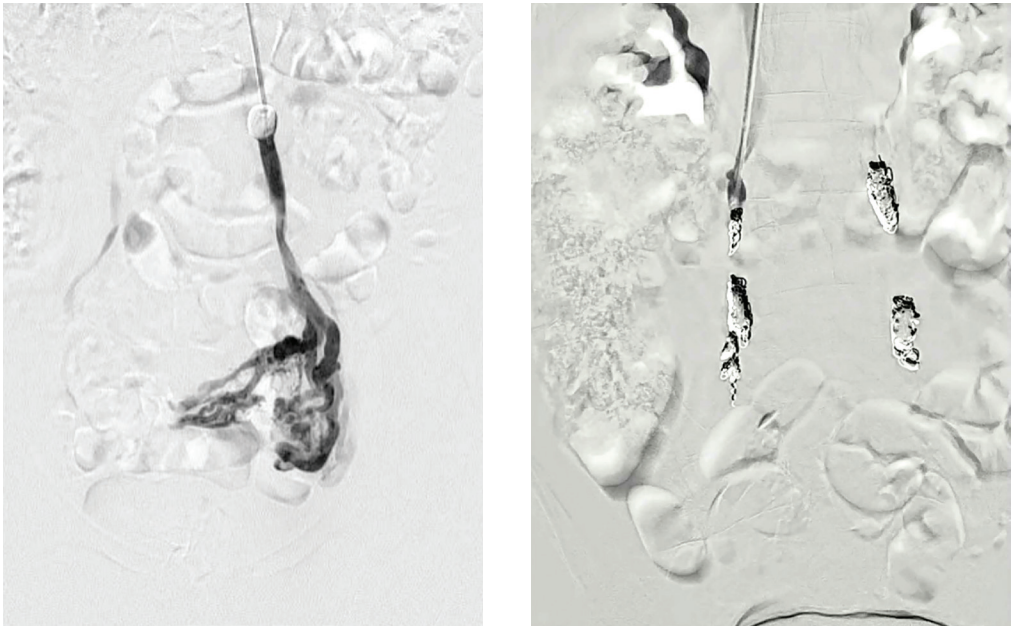
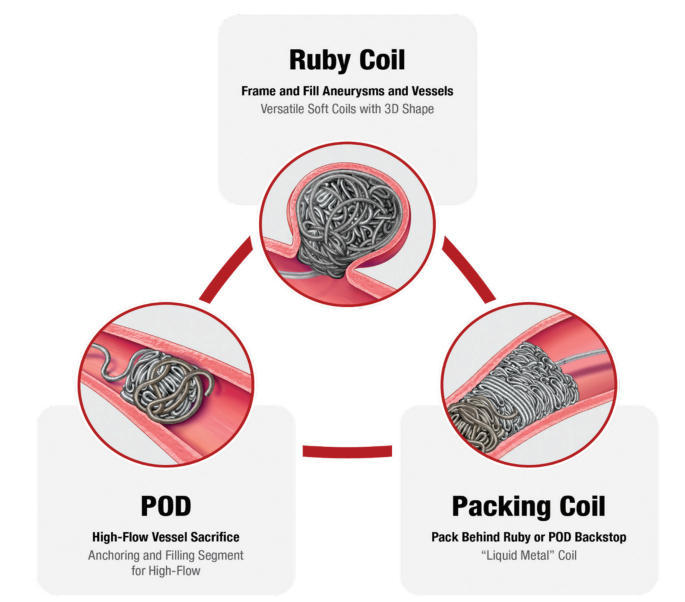
The Ruby, POD, and packing coils are all unique coils for different applications (aneurysm, vessel sacrifice, or space filling) and all designed to be large volume coils to reduce number of implants and cost of the procedure (Figure, Table). We are pushing to use packing coils but are allowing for the use of a POD coil at the top or bottom of the vein for complete safety to prevent any movement. The packing coil, or liquid metal coil, is designed to effectively fill space; the coil is the softest coil Penumbra offers and allows the operator to fill the primary vessel plus the collaterals, achieving complete occlusion of the pelvic veins (see images). The POD is designed with an anchoring distal tip and a proximal portion that becomes smaller/softer for efficient packing. Ruby can be used, but we are not expecting the need for a framing coil, given the POD’s unique ability to anchor within the vessel.
As part of the trial, we are doing injections to look at closure rates. We’re going to pass the coils, inject, and show that it is occlusive.
If EMBOLIZE shows what you expect, how do you see pelvic vein embolization fitting into standard practice?
Prior studies show that embolization has a high rate of technical success and that roughly 75% of women see immediate and sustained relief in their symptoms.i The treatment also has a low rate of complications. To date, we have not directly compared women treated with ovarian vein embolization to those who received no treatment. By comparing a treatment arm to a control arm, we expect EMBOLIZE will provide evidence of ovarian vein embolization’s efficacy in resolving significant pelvic pain, resulting in a greater number of women having access to this therapy.
Procedural and operative techniques and considerations are illustrative examples from physician experience. Physicians’ treatment and technique decisions will vary based on their medical judgment. The clinical results presented herein are for informational purposes only, and may not be predictive for all patients. Individual results may vary depending on patient-specific attributes and other factors.
Caution: Federal (USA) law restricts these devices for sale by or on the order of a physician. Prior to use, please refer to the Instructions for Use (IFU) for complete product indications, contraindications, warnings, precautions, potential adverse events, and detailed instructions for use. For the complete Penumbra IFU Summary Statements, please visit www.peninc.info/risk. Please contact your local Penumbra representative for more information.
This interview is sponsored by Penumbra, Inc.