The OPN NC Balloon in Complex PCI: Expanding What’s Possible
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of Cath Lab Digest or HMP Global, their employees, and affiliates.
CLD talks with Khaldoon Alaswad, MD, FACC, FSCAI
Director, Chronic Total Occlusion (CTO) Program; Director, Cardiac Catheterization Lab
Division of Cardiology, Henry Ford Health, Detroit, Michigan
How has lesion complexity changed in your percutaneous coronary intervention (PCI) practice over the past several years?
 There has clearly been a significant increase in lesion complexity in the cath lab over the past decade, largely because our patient population is getting older. We don’t eliminate coronary artery disease with statins and other therapies — we delay its onset and slow its progression. As a result, we are now seeing older patients with more calcified arteries, more tortuous vessels, and more diffuse and severe disease than what we were treating in the 1990s and early 2000s.
There has clearly been a significant increase in lesion complexity in the cath lab over the past decade, largely because our patient population is getting older. We don’t eliminate coronary artery disease with statins and other therapies — we delay its onset and slow its progression. As a result, we are now seeing older patients with more calcified arteries, more tortuous vessels, and more diffuse and severe disease than what we were treating in the 1990s and early 2000s.
It’s an interesting phenomenon. I travel to the Middle East and other parts of the world to do cases, and the disease pattern is different. In places where there hasn’t been the same widespread use of statins and preventive therapy, I actually see less calcification and less complexity. Even in Europe, I don’t see the same degree of complexity. Here in the U.S., our patients are older and their disease has been more chronic and longstanding. The progression has been slowed, but not eliminated, by risk factor control. So we end up managing more calcified, more tortuous, and more diffuse coronary artery disease.
Has the current focus shifted from simply delivering a stent to focusing on true stent optimization?
Yes, absolutely. With the increasing complexity of coronary artery disease, proper lesion preparation has become essential. It’s no longer enough just to deliver a stent. We have to deliver the right-size stent and ensure adequate expansion, apposition, and large stent area. That means displacing plaque effectively to achieve a minimum stent area of about 90% of the reference vessel diameter, or an absolute minimum of 5.5 mm² by intravascular ultrasound (IVUS) and 4.7 to 4.8 mm² by optical coherence tomography (OCT).
In the past, when disease was less complex, the process was simpler: you ballooned, deployed a stent, and assumed the result was adequate. That is no longer the case. Now, a significant amount of work goes into not just delivering the stent, but optimizing it.
We realized this once we began routinely using intravascular imaging with IVUS and OCT. Imaging showed us that we were underexpanding stents far more often than we thought, which likely contributed to suboptimal outcomes. Vessels are more complex than they appear angiographically, imaging can reveal previously underappreciated calcium in up to 70% of cases, and about one-third of those lesions show severe calcium that directly changes lesion preparation and stent expansion strategy.1
So, to truly optimize stent results, you need to address the calcium. The paradigm has clearly shifted. It’s no longer balloon, stent, and post dilate. It’s balloon, prepare the vessel properly, deploy the stent, and then optimize it carefully. Only then can you feel confident about long-term outcomes.
How has intravascular imaging changed your understanding of calcium and stent deployment?
It has completely changed it. Imaging changes calcium modification or stent deployment strategy in about two-thirds of complex cases.2 They may choose a larger balloon, a larger stent, or decide to post-dilate when they originally thought it wasn’t necessary.
Imaging showed us that lesions are often more calcified and more narrowed than they appear on angiography. It also allows us to better characterize calcium, whether it is superficial or deep, whether it involves 270 or 360 degrees of the vessel, and whether it is a short or long calcified segment.
Once we can define those characteristics, we can choose the appropriate modification strategy, whether that is atherectomy, the OPN NC balloon (Worldwide Innovations & Technologies, Inc.), specialty balloons, intravascular lithotripsy (IVL), or another approach. Imaging has also helped us understand how these different calcium types affect stent expansion and long-term outcomes.
How have trials such as VICTORY and ShortCUT influenced your use of ultra-high-pressure balloons?
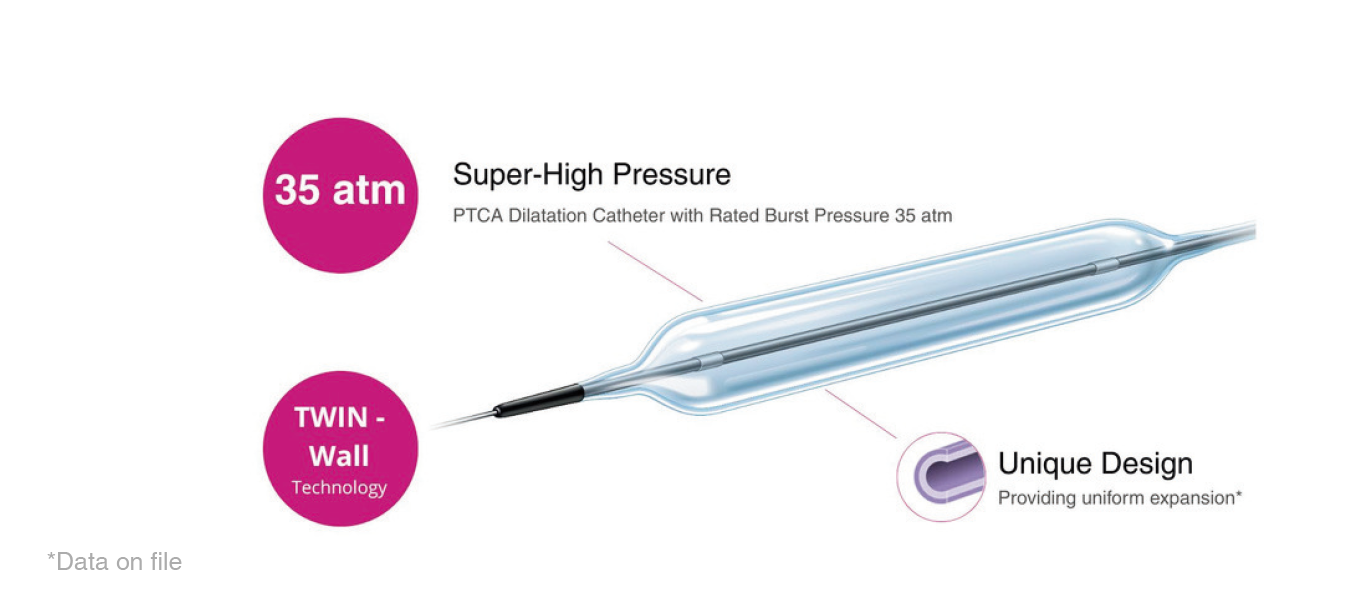
The VICTORY trial looked at OCT-guided PCI in severely calcified lesions and compared the OPN NC balloon with IVL. It showed that the OPN NC was non-inferior to IVL for final stent expansion (85% with OPN NC versus 84% with IVL). Procedural success was actually numerically higher with the OPN NC, 92.9% versus 86.1%, and strategy success without crossover was 98.6% in both groups.
Safety was comparable. Perforation rates were 1.4% with the OPN NC versus 3.6% with IVL, and 30-day outcomes like myocardial infarction and target vessel failure were similar. So when we talk about optimal stent expansion in calcified disease, we now have level-one randomized data showing that a super-high-pressure balloon can achieve the same result as IVL in complex lesions.

Another trial, ShortCUT, showed something similar, but with cutting balloons. In IVUS-guided PCI, cutting balloon angioplasty was non-inferior to IVL for achieving minimal stent area after rotational atherectomy. There were no differences in 30-day major adverse cardiovascular events, and balloon use came at a significantly lower device cost. So balloon-based plaque modification strategies are proving that they can stand next to IVL in appropriately selected cases.
VICTORY and ShortCUT reinforced what we were already seeing in practice. One of the big problems we had was in-stent restenosis. If operators didn’t use intravascular imaging and didn’t properly prepare the calcium, they often deployed a stent that was actually underexpanded. The patient then returns with in-stent restenosis, which is especially problematic if there are two layers of stent. Honestly, once you have in-stent restenosis with an underexpanded stent, it is very difficult to treat. We tried atherectomy and IVL, especially in two stent layers. The OPN NC is excellent for this scenario and has impressive results. I’ve had cases where a 2.5 mm stent was placed in a proximal left anterior descending (LAD) coronary artery that was actually 4.5 mm. That stent cannot expand beyond 3.7 mm, but with the OPN NC balloon, I was able not only to expand the stent, but actually split it open and then place a new stent appropriate for the vessel size.
For de novo lesions, you can clearly see the calcium fracture and the expansion. The key thing to be careful about when using the OPN NC is balloon sizing. Sizing should be 0.5 to 1.0 mm smaller than the reference vessel to reduce the risk of perforation.
Even before the VICTORY study was reported, we had already changed our practice to use the OPN NC balloon. In calcified or underexpanded stents, the OPN NC is usually my next step after a failed non-compliant balloon. There is also an economic component. IVL is expensive. Economically, the OPN NC solved a lot of problems for us. Instead of using IVL, we are able to achieve similar results with the OPN NC.
The ShortCUT trial showed no difference between IVL and cutting balloon in terms of achieving optimal stent area. If you extrapolate from that, and then look at the VICTORY study, which compared IVL versus balloon therapy, it further solidified our experience that there is no major difference. So now we have two trials, ShortCUT and VICTORY, both showing equivalency and not demonstrating much difference between IVL, which supposedly delivers about 50 atmospheres (atm) of pressure, and two balloon-based therapies. In my view, the OPN NC is faster to use and less expensive, and achieves similar results, as seen in the VICTORY trial.
In cases of in-stent restenosis, how important is deliverability?
It is very important in in-stent restenosis, especially in small or underexpanded stents. The deliverability of the OPN NC balloon is still suboptimal, but we can usually deliver it if we have enough guide support.
In very complex disease, deliverability is a challenge across the board. The cutting balloon, IVL, and even the OPN NC balloon can all be difficult to deliver. Often you have to prep the vessel first, either with ballooning or with rotational atherectomy, to create a pathway for the more definitive therapy. And that step is not wasted. Rotational atherectomy often does not achieve full calcium fracture. It opens up a pilot channel in the calcium so we can deliver devices, but it does not necessarily break deep calcium.
For in-stent restenosis, you typically pre-dilate first and then use the OPN NC balloon. You really don’t need rotablation in most of these cases. Before the OPN NC balloon was available, the only option for these patients was rotablation, which is technically contraindicated according to the instructions for use (IFU), but we used it because we had no alternative. We also tried cutting balloons, which worked sometimes, but most of the time they did not. We used high-pressure balloons at 18 or 24 atm, and sometimes we took balloons to pressures they weren’t designed for. Balloons designed to rupture at 18 atm were being taken to 24 atm trying to achieve expansion.
The OPN NC has now become first-line therapy for underexpanded stents in my practice.

You were an early adopter of the OPN NC. When did you begin using it, and what led you to adopt it so early?
I started using the OPN NC as soon as it was approved in the United States in late January 2023, so we are coming into the third year. We were early adopters because I would go to Europe and participate in cases, and our colleagues there showed it to me. I remember thinking, “I want this.” The Europeans had it way before we did. In fact, even before the OPN NC was available here, I was advocating for it. I went to some of the larger device companies and tried to convince them to bring this balloon to the U.S.
Where does the OPN NC fit in your escalation strategy for resistant lesions?
My usual approach is to start with a regular non-compliant balloon. If that doesn’t achieve adequate expansion, I move to the OPN NC balloon. One of the advantages of the OPN NC is that you can inflate and deflate it as many times as you need. It’s not like IVL, where you have a fixed number of pulses, and once you have used most of them, you are done. With the OPN NC balloon, if you choose the right size, you can dilate the entire vessel as needed.
When a lesion resists standard ballooning, how do you choose between the OPN NC, IVL, and atherectomy?
Honestly, a lot of it comes from habit and experience. We used to rely heavily on rotational atherectomy, and we still use it often. If I think the OPN NC or IVL may not deliver, I’ll start with rotablation to open a channel. Most of the time, that alone is adequate.
If it’s not adequate, meaning the lesion still does not expand on IVUS or OCT, then I will reach for the OPN NC first, not IVL. That’s generally how I approach it.
On the other hand, if I feel confident that I can deliver the OPN NC and I believe a regular non-compliant balloon won’t work, I’ll use the OPN NC upfront before the other options. There are several reasons for that: ease of use, familiarity, and practicality. It’s still just a balloon. There is no console, no plug, no additional equipment to set up. And there are also economic considerations.
So the decision really depends on deliverability, imaging findings, and what I think will be most efficient and effective in that particular lesion.
Can you walk us through your inflation technique and strategy with the OPN NC?
The IFU states 35 atm, and inflation should be done in stages. I typically inflate to 20 atm and count to five seconds, then go to 25 and count to five, then 30 and count to five, and then 35 and count to five.
Now, depending on how the lesion responds, if it doesn’t respond adequately at 35, I will go higher, up to 45 atm, and sometimes even 50 if needed.
In terms of duration, I hold each step for about five seconds. So if I go from 20 to 50 atm in five-atmosphere increments, holding each for five seconds, that ends up being about 30 seconds total inflation time.
Can you share a case that shows the utility of the OPN NC balloon?
Absolutely. Just last week, I had a patient with severe stenosis in the proximal left circumflex. The patient was actually referred to me for in-stent restenosis in the right coronary artery (RCA) after the referring physician had stented the proximal circumflex.
On imaging, we found severe in-stent restenosis in the circumflex. The vessel itself was a 4.5 to 5.0 mm vessel, but a 3.0 mm stent had been placed. When we performed IVUS, the stent was only expanded to about 2.0 mm in diameter at certain points.
We first tried to address it with a regular 3.5 mm × 20 mm non-compliant balloon. I intentionally used a longer balloon to avoid watermelon seeding, but the stent recoiled immediately. That’s when we escalated to a 4.0 mm OPN NC. As soon as we inflated it, I could see the stent open up and achieve much better expansion.
However, I couldn’t deliver the 4.0 mm OPN NC distally where there was another waist. So I switched to a 3.5 mm OPN NC, delivered it successfully, and inflated it to 40 atm. The distal waist completely disappeared. That segment had not responded to a regular non-compliant balloon at 25 to 26 atm.
In total, I used two OPN NC balloons and was able to fully expand the stent. This was a 3.0 mm stent that we ultimately expanded to above 4.0 mm. If it had been a 2.5 mm stent that couldn’t expand adequately, I would have had to split it open, which I have done before in the proximal LAD.
The stent had been placed less than three months earlier, so this was early restenosis. I did not need to place another stent because this was clearly a mechanical cause — and imaging studies suggest mechanical factors account for roughly 40–50% of ISR cases, particularly in early presentations.3 By restoring the stent to essentially the original vessel size, we created sufficient lumen area. Even if the patient develops intimal hyperplasia, it won’t be enough to re-occlude that space.
We treated the circumflex and stopped there. The patient’s RCA has now progressed to chronic occlusion, as the in-stent restenosis evolved into a chronic total occlusion. When we bring him back, we will likely use OPN NC again to prep the entire vessel, place a new stent in the de novo lesion, and probably use a drug-coated balloon for the non–de novo lesion.
In this case, the referring interventional cardiologist was able to deliver the stent to the proximal RCA but had to leave the distal lesion untreated because the lesion was resistant and needed extensive preparation that was not available locally. We’ll go back, prep the vessel, and complete the revascularization.
What has your experience been with the safety of ultra-high-pressure balloons?
Based on both trial data and our experience, the balloon has been safe. Even in the VICTORY study, there were low complication rates. If you look at the actual numbers, perforation was 1.4% with the OPN NC versus 3.6% with IVL, so super-high pressure did not translate into excess perforation risk in that trial.
In our anecdotal experience here, the OPN NC has also been safe. Now, there are certain situations that concern me. For example, if there is nodular calcium, where a pinhole could potentially be poked in the balloon. That would be concerning.
I have heard of balloon rupture causing problems, but personally, I have not had any complications, even in cases where the balloon ruptured.
How do you determine appropriate OPN NC sizing in de novo lesions versus in-stent restenosis?
I typically size the balloon 0.5 to 1.0 mm below the reference vessel. In a stented segment, I feel more emboldened because the stent creates scarring, especially if there are two layers of stent. In that case, I am comfortable going about 0.5 mm smaller than the reference vessel. But in a de novo lesion, I am more cautious and usually size closer to 1.0 mm smaller, relative to the distal reference vessel, not the proximal reference.
How do you see ultra-high-pressure balloon technology evolving over the next several years?
I think ultra-high-pressure balloons are going to see increasing use. For complex calcified PCI, OPN has become an essential tool in my lab, but we need to improve deliverability. The company is already working on making the OPN NC balloon more deliverable, especially through 6 French guides. And who knows, someone may eventually combine technologies. You could imagine an OPN NC with a drug coating, essentially an OPN NC plus DCB. Or combinations like OPN NC with IVL, or even integrating cutting balloon concepts. That way, if one technology doesn’t work, you have another mechanism built into the same platform. The sky is the limit in terms of innovation.
This interview is sponsored by Worldwide Innovations & Technologies, Inc.
References
1. Wang X, Matsumura M, Mintz GS, et al. In vivo calcium detection by comparing optical coherence tomography, intravascular ultrasound, and angiography. JACC Cardiovasc Imaging. 2017 Aug; 10(8): 869-879. doi:10.1016/j.jcmg.2017.05.014
2. Bergmark B, Dallan LAP, Pereira GTR, et al; LightLab Initiative Investigators. Decision-making during percutaneous coronary intervention guided by optical coherence tomography: insights from the LightLab Initiative. Circ Cardiovasc Interv. 2022 Nov; 15(11): 872-881. doi:10.1161/CIRCINTERVENTIONS.122.011851
3. Souteyrand G, Mouyen T, Honton B, et al. Stent underexpansion Is an underestimated cause of intrastent restenosis: insights from RESTO registry. J Am Heart Assoc. 2024 Nov 5; 13(21): e036065. doi:10.1161/JAHA.124.036065