Anatomical Approach to Atrial Leadless Pacing in the Region of Bachmann’s Bundle
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of EP Lab Digest or HMP Global, their employees, and affiliates.
EP LAB DIGEST. 2026;26(4):13-15.
Peter Farjo, MD, MS; Ashraf Alzahrani, MBBCh; Paari Dominic, MBBS, MPH; and Steven Bailin, MD
Division of Cardiovascular Medicine, University of Iowa Health Care, Iowa City, Iowa
Over the past decade, the development of leadless pacing systems has transformed the landscape of cardiac rhythm management. By eliminating the need for a surgical pocket and transvenous leads, these devices have reduced several well-recognized complications associated with conventional pacemakers, including infection, venous obstruction, and lead malfunction and dislodgement.1 Although their early use was limited to right ventricular pacing, ongoing technological advances now allow for atrial pacing and dual-chamber functionality.2
Traditional right atrial appendage (RAA) pacing, while technically straightforward, can introduce interatrial conduction delays and potentially predispose patients to atrial fibrillation (AF).3 In contrast, targeting Bachmann’s Bundle (BB), the preferential interatrial conduction pathway situated along the superior atrial septum, has been associated with shorter P-wave duration and improved atrial synchrony.4-7 While most data supporting BB pacing derive from transvenous systems, the possibility of combining this physiologic approach with leadless atrial technology presents a novel opportunity.
This report describes the implantation of a leadless atrial pacemaker in the region of BB, emphasizing procedural considerations, electrocardiographic findings, and short-term outcomes.
Case Presentation
Clinical History and Objective Data
A 68-year-old woman with a history of hypertension, type 2 diabetes mellitus, hyperlipidemia, obstructive sleep apnea, carotid artery disease, and prior left-sided invasive ductal breast carcinoma (ER/PR+, HER2–) presented with recurrent symptomatic sinus bradycardia. The patient had undergone lumpectomy followed by chemoradiation in 2022 and was under ongoing surveillance with serial breast magnetic resonance imaging (MRI).
In the last year, there were increasing symptoms of fatigue, lightheadedness, and 7 episodes of syncope, one of which occurred with a documented heart rate in the 40s.
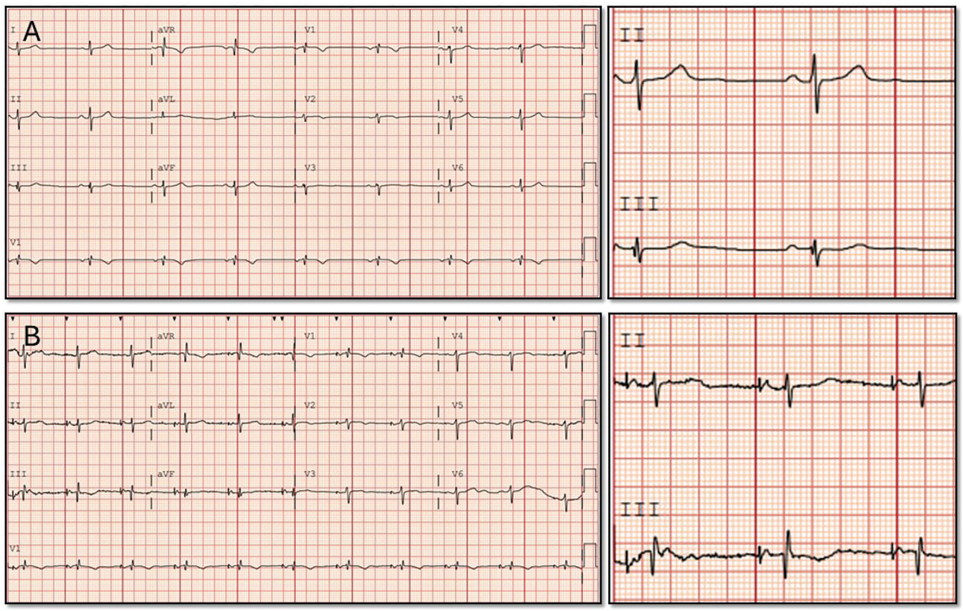
A 14-day Holter monitor revealed a predominant sinus bradycardia, with an average heart rate of 59 beats per minute (bpm) (range 41-114 bpm), 12 symptomatic triggers correlating with sinus bradycardia, and no significant atrioventricular (AV) nodal dysfunction. Echocardiography demonstrated a preserved left ventricular systolic function with an ejection fraction of 64%, normal atrial dimensions, and no significant valvular disease. Baseline electrocardiogram (ECG) showed sinus bradycardia at 54 bpm (Figure 1).
Because of symptomatic sinus node dysfunction, preserved AV conduction, and the clinical need for MRI compatibility, the patient was considered an appropriate candidate for implantation of a single-chamber AVEIR AR atrial leadless pacemaker (Abbott).
Procedural Description
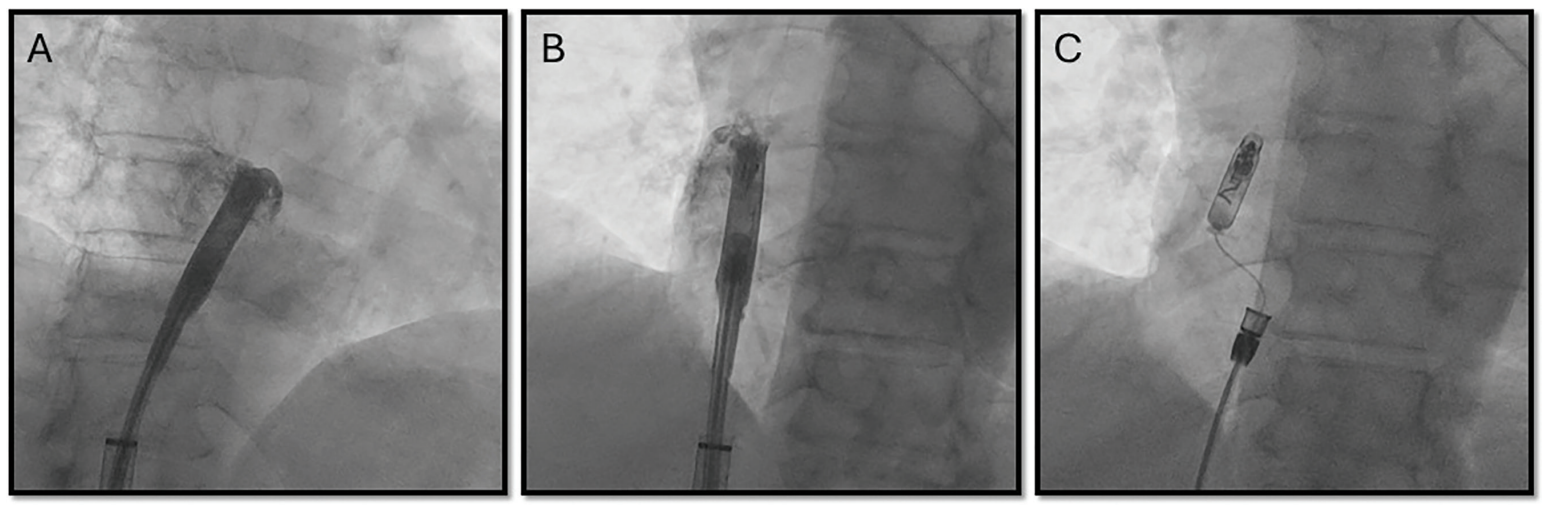
The procedure was performed in a cardiac electrophysiology laboratory under fluoroscopic guidance. Transfemoral venous access was obtained under sterile conditions, and the leadless pacemaker delivery system was advanced into the RA. To target the BB region, the system was advanced into the superior vena cava with the protected sleeve over the device. It was then withdrawn with anteroseptal deflection until the tip shifted toward the septal base of the RAA. The device was
then advanced under left anterior oblique (LAO) fluoroscopic view to direct the pacemaker toward the superior interatrial septum until the tip straightened along the RA roof, corresponding to the anatomical confluence of the interatrial septum and RA roof. Anterior positioning was confirmed in the right anterior oblique (RAO) view. Intracardiac contrast was injected in both views to verify localization at the anterior septal region (Figure 2) and mapping was performed prior to fixation displaying rapid P-wave upstrokes in leads II and III. Fixation was performed by screwing in the device with a half-body turn under anteroseptal deflection with the break. Once the torque of the half turn was fully conducted, the break was released while the device remained stable, and the remainder of the 1.5 total body turns were completed. Stability was confirmed by a tug test. Acute pacing and sensing thresholds were satisfactory, with atrial capture at 1.25 V @ 0.4 ms, impedance of 330 ohms, and sensing amplitude of 3.4 mV. Once parameters remained stable and fixation was confirmed, the tether was released and the delivery tool was withdrawn. Final fluoroscopic images in both LAO and RAO projections confirmed device placement at the junction of the RA roof and interatrial septum. The total procedure time was 36 minutes, with a fluoroscopy time of 5.3 minutes and radiation dose of 34.1 mGy.
Electrocardiographic Findings
Comparison of surface ECGs before and after implantation demonstrated electrocardiographic evidence of BB capture. P-wave duration shortened from 120 ms in sinus rhythm to 107 ms during pacing (Figure 3). The paced P-wave morphology showed brisk upright deflections in leads I, II, III, and aVF, with biphasic morphology in V1 and a reduction in P-wave morphology ≥10 ms, consistent with BB capture criteria.4,8-10
Follow-Up
At the 3-month interrogation, atrial capture threshold remained stable at 0.5 V @ 0.2 ms, with impedance of 290 ohms and sensing amplitude of 4.8 mV. Atrial pacing burden was 54%, and estimated device longevity was 6.7 years. No complications were noted, and the patient reported marked improvement in fatigue and lightheadedness, with no subsequent syncopal episodes.
Discussion
This case highlights the technical feasibility and early safety of delivering atrial pacing from the BB region using a leadless platform. Using fluoroscopic landmarks and electrocardiographic confirmation, the device was positioned in the superior interatrial septum. The final site demonstrated stable pacing parameters and physiologic P-wave characteristics. These findings suggest that leadless systems may be capable of replicating the results of transvenous BB pacing.
The concept of BB pacing stems from observations that conventional RAA pacing can prolong interatrial conduction and predispose to AF. Previous studies have shown that BB pacing produced shorter P-wave duration and was associated with a lower incidence and progression of AF compared with RAA pacing.4 In this case, P-wave duration decreased from 120 ms at baseline sinus rhythm to 107 ms during pacing, consistent with physiologic interatrial activation. However, not all studies have confirmed long-term clinical benefit; a meta-analysis found no difference in AF outcomes between septal and RAA pacing.7 This may reflect variability in patient populations, implant techniques, and definitions of BB capture.
Although this implant relied solely on fluoroscopic landmarks, more advanced approaches to BB localization have been described. For example, Lustgarten et al11 identified electrogram signatures characteristic of BB capture, yet noted that fluoroscopic placement alone achieves this in only about half of cases. Similarly, a recent report demonstrated leadless atrial implantation guided by intracardiac echocardiography and electrogram mapping, with documented improvements in conduction timing.12 While this approach illustrates that fluoroscopic targeting is reproducible and practical, it lacks the precision of electrogram-based guidance. Future iterations of leadless systems may need to integrate mapping capabilities to confirm physiologic capture.
Despite its promise, BB pacing using current leadless tools is not without challenges. Patient-specific atrial anatomy can complicate localization and maneuverability including a small volume RA or an aberrant inferior vena cava-RA angle, and fixation may be less predictable compared to more trabeculated sites such as the lateral RAA base. The absence of real-time electrogram feedback increases the risk of nonphysiologic pacing despite seemingly correct fluoroscopic placement. Additional procedural considerations include potential increases in radiation exposure, manipulation time, and the rare but serious risks of perforation or aortic erosion when engaging the atrial roof. While this case was uncomplicated, these theoretical risks should be acknowledged.
Summary
This case describes early adoption of leadless atrial pacing directed at the BB region, demonstrating its feasibility. Future work is warranted to validate and replicate this procedure in larger cohorts.
Disclosures: The authors have completed and returned the ICMJE Form for Disclosure of Potential Conflicts of Interest. Drs Farjo and Dominic report consulting fees from Boston Scientific and Biosense Webster; all payments are made to the University of Iowa. Dr Alzahrani has no conflicts of interest to report. Dr Bailin reports consulting fees from Abbott and Medtronic, as well as consulting fees from Boston Scientific to the institution.
References
- Piccini JP, El-Chami M, Wherry K, et al. Contemporaneous comparison of outcomes among patients implanted with a leadless vs transvenous single-chamber ventricular pacemaker. JAMA Cardiol. 2021;6(10):1187-1195. doi:10.1001/jamacardio.2021.2621
- Knops RE, Reddy VY, Ip JE, et al. A dual-chamber leadless pacemaker. N Engl J Med. 2023;388(25):2360-2370. doi:10.1056/NEJMoa2300080
- Cronin EM, Vedage N, Israel CW. Alternative atrial pacing site to improve cardiac function: focus on Bachmann’s bundle pacing. Eur Heart J Suppl. 2023;25(Supplement_G):G44-G55. doi:10.1093/eurheartjsupp/suad118
- Bailin SJ, Adler S, Giudici M. Prevention of chronic atrial fibrillation by pacing in the region of Bachmann’s Bundle: results of a multicenter randomized trial. J Cardiovasc Electrophysiol. 2001;12(8):912-917. doi:10.1046/j.1540-8167.2001.00912.x
- Verlato R, Botto GL, Massa R, et al. Efficacy of low interatrial septum and right atrial appendage pacing for prevention of permanent atrial fibrillation in patients with sinus node disease: results from the Electrophysiology-Guided Pacing Site Selection (EPASS) study. Circ Arrhythm Electrophysiol. 2011;4(6):844-850. doi:10.1161/circep.110.957126
- Padeletti L, Pieragnoli P, Ciapetti C, et al. Randomized crossover comparison of right atrial appendage pacing versus interatrial septum pacing for prevention of paroxysmal atrial fibrillation in patients with sinus bradycardia. Am Heart J. 2001;142(6):1047-1055. doi:10.1067/mhj.2001.119373
- Shali S, Su Y, Ge J. Interatrial septal pacing to suppress atrial fibrillation in patients with dual chamber pacemakers: a meta-analysis of randomized, controlled trials. Int J Cardiol. 2016;219:421-427. doi:10.1016/j.ijcard.2016.06.093
- MacLean WA, Karp RB, Kouchoukos NT, James TN, Waldo AL. P waves during ectopic atrial rhythms in man: a study utilizing atrial pacing with fixed electrodes. Circulation. 1975;52(3):426-434. doi:10.1161/01.cir.52.3.426
- Das A, Banerjee S, Mandal SC. A simple method for Bachmann’s bundle pacing with indigenous modification of J-stylet. Indian Heart J. 2016;68(5):678-684. doi:10.1016/j.ihj.2015.10.299
- Infeld M, Habel N, Wahlberg K, et al. Bachmann bundle potential during atrial lead placement: a case series. Heart Rhythm. 2022;19(3):490-494. doi:10.1016/j.hrthm.2021.11.015
- Lustgarten DL, Habel N, Sánchez-Quintana D, et al. Bachmann bundle pacing. Heart Rhythm. 2024;21(9):1711-1717. doi:10.1016/j.hrthm.2024.03.1786
- Hollis Z, Yang E, Ryu K, et al. Leadless atrial pacing targeting Bachmann’s Bundle for atrial resynchronization. JACC Case Rep. 2025;30(20):104083. doi:10.1016/j.jaccas.2025.104083










