Adopting Pulsed Field Ablation: Visualization, Catheter Management, and Fluoroless Workflow Strategies
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of EP Lab Digest or HMP Global, their employees, and affiliates.
EP LAB DIGEST. 2026;26(4):9-12.
Interview by Jodie Elrod
As electrophysiology (EP) programs expand in both size and procedural complexity, the ability to maintain safety, efficiency, and procedural effectiveness becomes increasingly important. In this interview, Sri Sundaram, MD, Co-Director of the National Cardiac EP Service Line, and Chair EP, Rocky Mountain Division, AdventHealth, shares insights from a high-volume program performing more than 3000 EP procedures and approximately 1000 ablations annually, nearly all without fluoroscopy. The discussion highlights key considerations in adopting pulsed field ablation (PFA) technologies, implementing a fully fluoroless workflow, and optimizing visualization and catheter management to support consistent outcomes in everyday clinical practice.
Can you provide an overview of the EP program at AdventHealth Littleton and Porter, including annual procedural volume and the key technologies in use?
 We just reviewed our year-end numbers, and between the 2 sites, we perform a little more than 3000 EP procedures combined, with about 1000 ablations per year. When we look at our volumes, we are seeing year-over-year growth of more than 20%, and that has been consistent annually. A lot of this growth is attributable to 2 things: first, we believe we are a very strong program that continues to expand, and second, Colorado’s population growth has made a meaningful impact as well.
We just reviewed our year-end numbers, and between the 2 sites, we perform a little more than 3000 EP procedures combined, with about 1000 ablations per year. When we look at our volumes, we are seeing year-over-year growth of more than 20%, and that has been consistent annually. A lot of this growth is attributable to 2 things: first, we believe we are a very strong program that continues to expand, and second, Colorado’s population growth has made a meaningful impact as well.
As a growing program, we now consider ourselves high volume. We want access to all available technologies. My goal as EP section head is to ensure that my partners and I have everything we need to tailor treatments for our patients and to use whatever technology we believe is best for them. Because of that, we tend to have them all. Currently, we are using the Affera™ mapping and ablation system (Medtronic), Volt PFA system (Abbott), and Farapulse PFA platform (Boston Scientific).
What key factors or considerations have guided your decision-making when selecting PFA technologies for your practice?
Safety is the primary consideration—it must always center on patient outcomes. We reviewed clinical trials, evaluated complication rates, and assessed what was both realistic and reproducible in practice, with the goal of maintaining consistently low complication rates.
Efficacy is the next key factor. We prioritize systems that demonstrate reliable clinical performance. In reviewing the Affera mapping and ablation system trial data, the platform showed comparatively high 1-year procedural success rates. Based on these results, we sought hands-on evaluation to better assess its performance.
At Heart Rhythm 2025, the Medtronic team facilitated access to the technology booth, where my partners and I were able to test the system directly. The experience demonstrated a high degree of flexibility, ease of use, and intuitive design. Based on this evaluation, it became clear that this technology would play a central role in our practice over the coming years.

Can you describe your workflow when using the Sphere-9™ catheter with the Affera mapping and ablation system, with a specific focus on your visualization strategies?
I have been fluoroless for more than 10 years. With the exception of one physician who uses approximately a minute of fluoroscopy, my entire group is fluoroless as well. As a result, we perform essentially all 1000 ablations per year without fluoroscopy.
The transition to a fluoroless workflow with the Affera mapping and ablation system was not difficult. Our workflow begins with placement of the intracardiac echocardiography (ICE) catheter into the right atrium (RA), followed by a comprehensive survey to confirm that cardiac anatomy appears as expected and that there are no unexpected findings. We then proceed with the transseptal puncture. Most of us now perform this using only an SL1 and a BRK, without relying on more advanced or higher-cost sheaths.
After transseptal access, we create a left atrial (LA) map using the Affera mapping and ablation system. In my own practice, I have increasingly placed the ICE catheter in the LA, which has become standard for most of my cases. I find that this approach provides even better visualization of the Affera mapping and ablation system compared with imaging from the RA, and this has become my preferred workflow.
There are some cases in which an ICE catheter does not easily advance into the RA. In those situations, while it is possible to obtain acceptable visualization from the RA, the image quality is not optimal.
What features of the Sphere-9 catheter make it well-suited to support your low-fluoroscopy procedures?
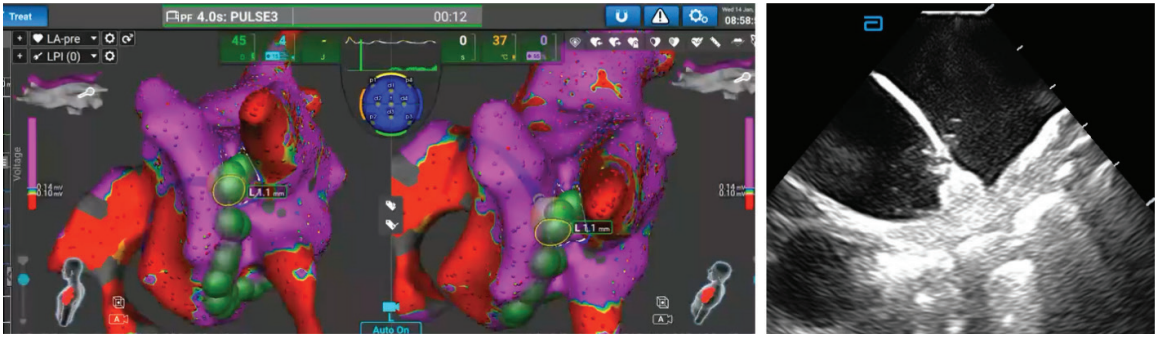
The Sphere-9 catheter is highly visible on echocardiography. It can be seen very clearly, which makes a significant difference, particularly for operators who are not heavy ICE users or for those working toward low or minimal fluoroscopy workflows. The ability to clearly visualize the ablation catheter on ICE is extremely valuable. It is highly echo reflective, and that visibility meaningfully improves procedural confidence and efficiency.
In addition, the catheter allows mapping, pacing, and ablation, enabling all necessary functions to be performed from a single catheter. This eliminates the need for a second catheter in the LA to complete the procedure. As a result, there is no concern about having 2 catheters in the LA or about potential interactions on ICE, where one catheter can obscure the other and limit visualization. Using a single catheter simplifies the workflow. Combined with its strong echo reflectivity on ICE, this streamlined approach is what makes the system particularly easy to use.
What features of the Affera mapping and ablation system and Prism-2 software complement the Sphere-9 catheter in facilitating a low-fluoroscopy workflow?
Visualization of other catheters is also important, even though the entire procedure can be performed with the Sphere-9 catheter. At certain points, additional catheters are still helpful. For example, it is often useful to pace from another catheter when needed, most commonly a coronary sinus (CS) catheter, which is what I typically use. This also provides added stability if the patient becomes bradycardic, since pacing can be performed as needed. In cases of atypical flutter, the CS catheter is helpful for assessing directionality and cycle length, which are easier to interpret with this additional reference.
What the Prism-2 software has done is make placement of the CS catheter significantly easier. Previously, I relied entirely on ICE for this step, and even with my level of comfort using ICE, it would still take approximately 4 to 5 minutes. With Prism-2 software, I can now complete the placement in less than a minute. In addition, I am able to visualize the catheter throughout the case to confirm that it remains stable. If it becomes dislodged or migrates to a different location, I can immediately recognize that change without concern.
Based on your experience, what insights or practical advice would you share with electrophysiologists looking to implement the Sphere-9 catheter and the Affera mapping and ablation system for low- or no-fluoroscopy procedures?
The Sphere-9 catheter is extremely easy to use. I began performing ablations during the point-by-point era, and even going point by point, the system is very straightforward. The reach is substantial, with a 9-millimeter design, which is reflected in the name Sphere-9, and this makes catheter manipulation much easier. The catheter is highly flexible, making it accessible for a wide range of operators.
The ability to map, pace, and ablate with a single catheter further simplifies the workflow. All necessary functions can be performed without catheter exchanges, which adds to procedural efficiency. From a low-fluoroscopy perspective, the 8 French profile also contributes to ease of use, as there is no concern about a large sheath interfering with ICE imaging.
Overall, the catheter is highly visible and intuitive to use. It is an accessible piece of equipment that can be readily adopted by most operators. The learning curve is minimal, particularly for those with prior experience performing point-by-point ablation, since the fundamental workflow is already familiar.
The transcripts were edited for clarity and length.
Disclosures: Dr Sundaram has completed and returned the ICMJE Form for Disclosure of Potential Conflicts of Interest. He reports consulting fees from Medtronic, and payment or honoraria for speaker’s bureaus.
This content was published with support from Medtronic.










