Is a Mapping System Needed for Pulsed Field Ablation Cases?
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of EP Lab Digest or HMP Global, their employees, and affiliates.
EP LAB DIGEST. 2026;26(4):6.
Bradley P Knight, MD, FACC, FHRS
Dear Readers,
A 69-year-old woman with recurrent persistent atrial fibrillation (AF) developed recurrent AF and atypical atrial flutter after 4 catheter ablation procedures elsewhere over the past 10 years. These procedures included pulmonary vein isolation (PVI), ablation of the cavotricuspid isthmus (CTI), and additional focal atrial ablation. Her most recent ablation procedure was performed over a year ago. During that procedure, redo PVI and ablation of the posterior left atrium (PLA) was reportedly performed using a pentaspline pulsed field ablation (PFA) catheter (Farawave, Boston Scientific) and a nonintegrated 3D mapping system.
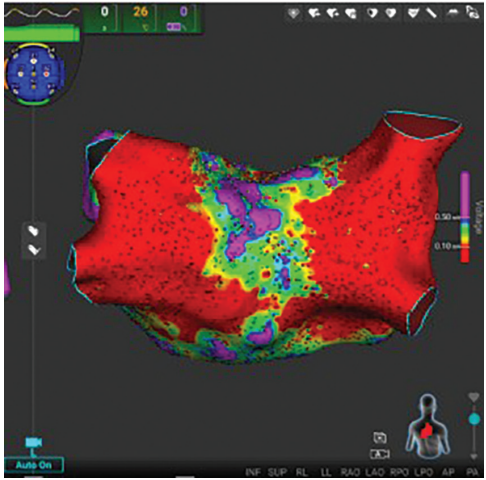
The patient presented for her fifth ablation procedure in sinus rhythm. An Affera 3D Mapping and Ablation System and a Sphere-9 dual-energy PF/RF catheter (Medtronic) were used for mapping and ablation. A voltage map was made of the LA (Figure). Surprisingly, there were extensive areas of incomplete ablation of the PLA. How could that be?
Further investigation of her prior procedure revealed that a 3D mapping system was used, but only during the PVI portion. The nonintegrated 3D mapping system reportedly became nonoperational toward the end of the procedure, when ablation of the PLA was performed. As a result, ablation of the PLA was performed with the pentaspline catheter in the flower configuration guided by fluoroscopy alone.
In the United States, use of a 3D mapping system during ablation procedures for AF is routine. However, in Europe—partly due to cost constraints—PVI is often performed using fluoroscopy alone, as has long been done with cryoballoon technology.
To evaluate whether a mapping system is necessary for PFA, a group in Basel, Switzerland, studied the role of 3D electroanatomic mapping on procedural characteristics and outcomes in patients undergoing PFA for AF.1 They compared patients undergoing PVI only with a 31-millimeter Farawave (Boston Scientific) PFA catheter. They compared 127 patients who underwent PVI with a mapping system and 70 patients with no mapping. Baseline characteristics were similar between groups.
Use of a 3D mapping system was associated with substantial increases in procedure times, with the LA dwell time more than doubling when a mapping system was used. Acute PVI success was 100% in both groups. During a median follow-up of 267 days, atrial arrhythmia recurrence occurred in 14% of the mapping group and 17% of the non-mapping group, a difference that was not statistically significant. The authors concluded that there was no objective added benefit to using 3D mapping for PFA-guided PVI.
In the present case, reconnection of the PLA may have been due to reversible PFA lesions created with the pentaspline catheter. However, it seems more likely that areas of nonablated tissue persisted because portions of the PLA were never actually ablated when the procedure was performed under fluoroscopic guidance alone.
Despite the findings of the European study, this case illustrates that a 3D mapping system can provide meaningful value during AF ablation with PFA. It may be that the value is limited for PVI alone, but when additional lesions—including ablation of the entire PLA—are performed, a mapping system is useful. In this patient, treatment consisted of targeted ablation of the posterior wall and the anterior mitral isthmus with the Sphere-9 catheter, addressing both AF and spontaneously occurring perimitral flutter.
Disclosures: Dr Knight has served as a paid consultant to Medtronic and was an investigator in the PULSED AF trial. He has served as a consultant, speaker, investigator, and/or has received EP fellowship grant support from Abbott, AltaThera, AtriCure, Baylis Medical, Biosense Webster, Biotronik, Boston Scientific, CVRx, Philips, and Sanofi; he has no equity or ownership in any of these companies. Dr Knight reports payment or honoraria from Convatec for a lecture.














