Integrating Intuition With Insight: Redesigning Cardiovascular Remote Monitoring at an Academic Medical Center
A white paper by RhythmScience
A white paper by RhythmScience
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of EP Lab Digest or HMP Global, their employees, and affiliates.
EP LAB DIGEST. 2026;26(4):16-17.
A white paper by RhythmScience
Background and Rationale
Academic medical centers face mounting pressure as cardiovascular care becomes increasingly data-intensive. At the University of Chicago Medicine (UCM), electrophysiology (EP), heart failure (HF), and remote monitoring (RM) teams were challenged by rapid growth in cardiac implantable electronic device (CIED) volumes, expanding physiologic data streams, staffing constraints, and the need to maintain timely and safe clinical response. These pressures prompted leadership to re-evaluate how cardiovascular data was managed, reviewed, and operationalized across the enterprise. This article explores the pre- and post-implementation experiences utilizing the Rhythm360 (RhythmScience) CIED and HF Patient Management solution. Interviews were conducted with:
 Gaurav A Upadhyay, MD, FACC, FHRS, Professor of Medicine and Director of the Pacing & Defibrillation Device Clinic. Dr Upadhyay is an electrophysiologist.
Gaurav A Upadhyay, MD, FACC, FHRS, Professor of Medicine and Director of the Pacing & Defibrillation Device Clinic. Dr Upadhyay is an electrophysiologist.
 Andrew Beaser, MD, Associate Professor of Medicine. Dr Beaser specializes in CIED device management.
Andrew Beaser, MD, Associate Professor of Medicine. Dr Beaser specializes in CIED device management.
 Lauren Bowles, MSN, FNP-BC, is a member of the Advanced Practice HF team.
Lauren Bowles, MSN, FNP-BC, is a member of the Advanced Practice HF team.
Before Implementation: Fragmented and Resource-Intensive Workflows
Prior to implementing Rhythm360, UCM relied on a fragmented monitoring environment. Device data was accessed through multiple OEM portals, requiring manual review, summarization, and physician sign-off. HF diagnostics, including pulmonary artery pressure monitoring (eg, CardioMEMS, Abbott), were managed separately, which limited cross-team visibility and increased documentation burden. This constrained scalability and heightened risk of delayed recognition of clinically meaningful events.
Dr Beaser described the workflow as “a major challenge and incredibly difficult.” Dr Upadhyay emphasized operational strain: “Staffing was always an issue for our center, because our device clinic—like many other medical centers—had struggled with technician turnover and timely weekend coverage.”
Implementation Approach: Redesigning the Monitoring Model
UCM selected Rhythm360 not simply as a technology platform, but as a full-service model for CIED, HF, and remote patient monitoring implementation designed to address workflow, staffing, and integration challenges.
Key components included:
- Centralized aggregation of multi-vendor device data
- Service-supported first-level review with physician oversight
- Epic integration to improve data accessibility
- Mobile tools aligned with clinician workflows
Staffing support was a decisive factor. As Dr Upadhyay stated, “That was a big piece for us, to have an integrated review of data from trained personnel.”
After Implementation: Operational and Clinical Impact
Following implementation, UCM observed meaningful changes in monitoring workflow. Clinicians were able to review more transmissions daily while improving timeliness of identifying actionable findings. Dr Beaser noted, “We are seeing more of these transmissions per day and identifying more abnormalities.”
Earlier recognition enabled proactive intervention. “We are able to address these issues earlier; rather than waiting for a 3-month visit, we can call patients in for evaluation,” Dr Beaser explained.
From a financial and operational perspective, improved integration supported more consistent billing processes. As Dr Upadhyay observed, “We have improved billing and accountability for our patients after the integration.”
Mobile access through the Rhythm360 physician application further aligned monitoring with clinical workflows. Dr Beaser highlighted its practical value, stating, “I am more likely to sign off on these while in meetings because I can easily access them on my phone.”
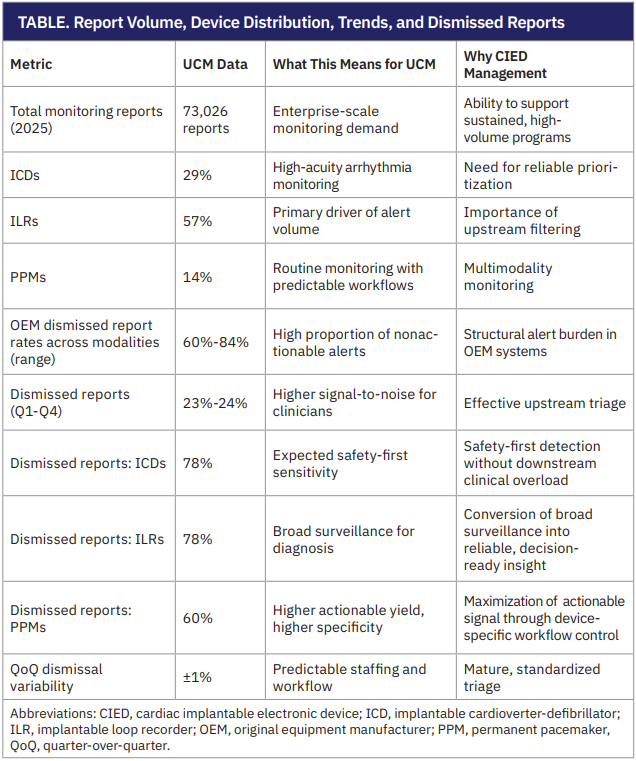
Operational and Clinical Context for Monitoring Metrics
To contextualize clinician experience, UCM analyzed aggregate monitoring data generated through Rhythm360 during calendar year 2025. The review included total report volume, device distribution, quarterly trends, and dismissed-report patterns (Table).
Together, they illustrate how centralized triage supports predictable staffing, sustained coverage, and clinical prioritization in an environment characterized by high data volume and limited tolerance for variability.
Lessons Learned and Future Direction
UCM’s experience highlights that high dismissal rates in CIED monitoring are not vendor-specific anomalies but structural features of safety-first RM. Across more than 73,000 reports reviewed annually by UCM, most OEM-generated alerts were nonactionable, reinforcing that clinical burden is driven less by true events than by the total number of device-generated alerts and transmissions requiring review (ie, signal volume). In this context, staffing models alone are insufficient to manage growth in monitoring demand.
Centralized, service-supported triage allowed UCM to manage consistently high report volume, averaging more than 18,000 reports per quarter, while keeping dismissal rates stable across device types and throughout the year. This consistency indicates that standardized triage criteria and workflow design can reduce day-to-day variability in clinical workload, making staffing and coverage needs predictable.
Looking forward, participants identified decision support and multimodality data integration as natural extensions of this model. As monitoring volume continues to increase, future efforts will likely focus on enhancing clinical prioritization through contextualized alerts, longitudinal trend analysis, and data integration across EP and HF programs, ensuring safety and scalability without increasing clinician burden.
The UCM experience also underscored the importance of selecting a partner capable of rapid iteration and responsiveness to clinician feedback. Early usability and integration challenges informed ongoing refinement, reinforcing the value of collaboration between clinical teams and technology partners.
Looking forward, clinicians identified opportunities to further enhance decision support and integrate multimodality data. As Dr Beaser noted, “Decision support, including AI-assisted decision support, will become increasingly important as data volumes grow.”
Conclusion
UCM’s implementation of a centralized, service-supported monitoring model demonstrates how large academic programs can sustainably manage the growing complexity of CIED and physiologic monitoring. Across more than 73,000 reports annually and consistently high quarterly volumes, UCM maintained stable triage and dismissal patterns, allowing clinicians to focus on reports most likely to require action while preserving patient safety.
By shifting report dismissal upstream and standardizing first-level review, UCM aligned staffing, workflow, and clinical oversight in a way that reduced downstream burden without compromising vigilance. This experience suggests that effective CIED management is less about reducing alert generation and more about designing systems that can absorb scale, variability, and signal noise. For organizations evaluating RM strategies, the UCM experience highlights the importance of workflow architecture, triage consistency, and multidisciplinary integration as foundational elements for long-term sustainability.
This content was published with support from RhythmScience.
Disclosures: The authors have completed and returned the ICMJE Form for Disclosure of Potential Conflicts of Interest. Outside this manuscript, Dr Upadhyay reports that he is a consultant on the RhythmScience scientific advisory board and serves as a consultant for Abbott, Biotronik, Boston Scientific, GE HealthCare, Medtronic, Philips, and Zoll; he also reports grants for studies from Abbott, Biotronik, Boston Scientific, and Medtronic. In addition, Dr Upadhyay reports speaking payments for Medtronic, Boston Scientific, and Zoll; he also reports support for travel from Abbott, Boston Scientific, Medtronic, and Philips. Dr Beaser and Ms Bowles have no conflicts of interest to report.










