Utilization of a Novel Porcine Extracellular Matrix as a Dermal Template for Skin Graft
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of ePlasty or HMP Global, their employees, and affiliates.
Abstract
Background. Achieving a vascularized granulation bed is essential prior to split-thickness skin grafting (STSG) but is often protracted with existing dermal substitutes. Porcine-derived extracellular matrix (pECM) powder ( XCelliStem Wound Powder; StemSys) supplies structural proteins and growth factors that may shorten this interval.
Methods. Three adults with complex wounds—a traumatic dorsal-foot defect, a cervical burn contracture reconstructed with a latissimus flap, and a free omental flap to an upper-extremity degloving injury—received intraoperative application of hydrated pECM powder following meticulous debridement. Wounds were covered with Xeroform and either hypochlorous-soaked gauze or negative pressure wound therapy. Granulation, exudate, and infection were assessed daily; STSG was performed once a uniform, vascular bed formed. Patients were followed for ≥3months.
Results. Complete, healthy granulation tissue developed on post-application days 9, 11, and 13, respectively, enabling timely STSG. All grafts achieved 100% take without necrosis, seroma, or infection. At 3 months, each site displayed durable coverage, preserved function, pliable scars with preserved elasticity, and high patient satisfaction.
Conclusions. In this preliminary series, pECM powder accelerated wound bed readiness and supported reliable STSG within 2 weeks—substantially faster than the 3- to 7-week intervals reported for Integra Bilayer Matrix (Integra LifeSciences Corp) and NovoSorb Biodegradable Temporizing Matrix (PolyNovo Ltd). The pECM powder appears safe, easy to apply, and may reduce morbidity and resource use; controlled trials are warranted.
Introduction
The successful application of split-thickness skin grafts (STSGs) in reconstructive surgery is dependent on the establishment of a well-vascularized, stable wound bed capable of supporting graft adherence and integration. Integral to this process is the formation of granulation tissue, serving as the biological scaffold between the wound bed and the overlying graft. While some surgeons advocate for immediate grafting over freshly debrided, nongranulated wounds in cases such as burns or trauma, this approach may carry a risk of poor adherence if residual contamination or inadequate vascularity is present.1-2 In contrast, the use of a granulating wound bed, especially in complex or previously contaminated wounds, may provide a more stable substrate for graft survival. Granulation tissue supports processes including cellular migration, angiogenesis, nutrient diffusion, and reepithelialization, all of which contribute to long-term graft survival and functional skin regeneration.1-4 However, achieving adequate granulation tissue in complex, large, or contaminated wounds remains a substantial clinical challenge. These wounds may demonstrate delayed healing trajectories secondary to impaired vascular supply, infection risk, and patient-specific factors such as diabetes or immunosuppression.5-6 Delays in wound bed preparation can prolong hospitalization and hinder adequate closure, in turn increasing the burden on health care resources and patient quality of life.
To address these issues, dermal regeneration templates such as Integra Bilayer Wound Matrix (BWM; Integra LifeSciences Corp) and NovoSorb Biodegradable Temporizing Matrix (BTM; PolyNovo Ltd) have been widely implemented. These synthetic or biosynthetic matrices act as temporary scaffolds, supporting neodermis formation prior to grafting. While they have been shown to demonstrate efficacy in promoting dermal regeneration, their integration timelines remain lengthy. Clinical studies report mean times to graft readiness ranging from approximately 21 to 30 days for BWM and 34 to 53 days for BTM.7-9 These extended durations to readiness are especially burdensome in critically ill patients, in which prolonged time to closure may promote increased rates of severe complications.
In response to the limitations of traditional dermal substitutes, biologically active wound healing agents have emerged as promising alternatives. Among them is XCelliStem Wound Powder (StemSys), a porcine spleen- and lung-derived acellular extracellular matrix (pECM) formulated as a sterile powder. Unlike traditional bilayer constructs, pECM powder can be applied directly to the wound surface, allowing it to conform to irregular wound geometries and facilitate direct contact with viable tissue. pECM powder delivers key structural proteins, including collagen (Types I, III, IV, V), laminin, fibronectin, glycosaminoglycan, and growth factors to the wound bed. These bioactive proteins, termed “matrikines” stimulate early cellular migration, angiogenesis, and matrix remodeling.10-13 They provide additional support for host cell infiltration, neovascularization, and the organized deposition of new collagen, ultimately facilitating a biologically integrated wound bed that can support autologous skin grafting. Histologic studies on similar porcine matrices have shown reduced scar formation and improved elasticity in deep burn wounds.14 Additionally, the ease of application and biocompatibility of pECM powder may offer a practical advantage in accelerating wound bed preparation even in patients with extensive comorbidities or complex injuries. These attributes may be particularly beneficial in acute care settings or resource-limited environments.
This case series presents 3 patients with complex wounds whereby pECM powder was utilized to accelerate wound bed readiness prior to STSG placement. The speed and quality of granulation tissue formation are evaluated relative to conventional dermal substitutes, offering insights into the potential efficiency and utility of pECM powder across diverse clinical scenarios.
Materials and Methods
Patient selection and clinical evaluation
This study included 2 patients with complex soft tissue injuries—1 traumatic and 1 burn related—who required accelerated wound bed preparation to facilitate timely skin grafting. Selection criteria required wounds with healthy, well-perfused tissue beds but delayed epithelialization or incomplete granulation, making them suitable for regenerative matrix therapy. Each patient was assessed for overall medical stability, with exclusion of those presenting with systemic infection, immunocompromised status (including corticosteroid therapy or active malignancy), or poorly controlled diabetes (HbA1c > 9.0%).
Surgical preparation and debridement
In all cases, surgical wound preparation was carried out in the operating room under sterile conditions. All nonviable soft tissue was removed via sharp excisional debridement until viable, bleeding tissue was visualized.
Application of regenerative matrix
Once the wound bed was determined to be free of infection and demonstrated a healthy vascular surface, pECM powder was applied. Prior to use, the powder was lightly hydrated with sterile normal saline (0.9%) to facilitate adhesion. It was then evenly distributed across the entire surface of the wound in a thin, uniform layer to ensure direct contact with the underlying tissue.
To maintain a moist wound environment and protect the matrix, a single layer of Xeroform gauze was placed directly over the pECM powder. This dressing was then secured circumferentially to the wound margins to prevent displacement of the matrix and maintain conformity to the wound bed.
Following fixation, a standard secondary dressing protocol was initiated. This included wet-to-dry dressing changes with a dilute hypochlorous acid solution (case 1 and 2) or negative pressure wound therapy (case 3).
Postoperative monitoring and skin grafting
Wounds were evaluated daily for signs of granulation tissue development, exudate level, and infection. Once a robust, vascularized granulation bed was observed—on postoperative day (POD) 11 in case 1, POD 9 in case 2, and POD 13 in case 3—a STSG was harvested from the anterolateral thigh.
Follow-up and outcome assessment
Postoperative graft monitoring was conducted both inpatient and outpatient. Graft success was assessed based on adherence, presence of necrosis, and infection. In both cases, photographic documentation was obtained at key milestones: matrix application, early graft incorporation, and follow-up evaluation. Long-term outcomes included graft durability, cosmetic appearance, and functional recovery, as determined by physical examination and patient-reported satisfaction. Each patient was followed for a minimum of 3 months after final surgical intervention.
Results
Three patients with complex wounds underwent wound bed preparation using pECM powder followed by STSG. All patients demonstrated favorable clinical outcomes, with rapid granulation tissue formation and successful graft adherence observed within a short timeframe.
Case 1: complex traumatic foot wound
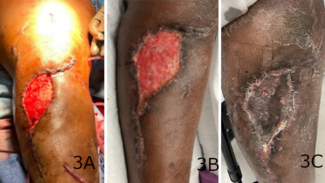
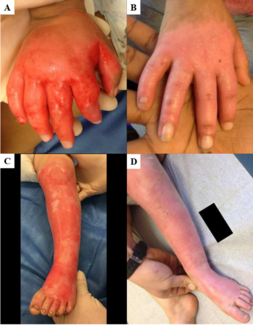
An 80-year-old male sustained a high-impact crush injury to his right foot, resulting in extensive soft tissue trauma (Figure 1). After initial surgical stabilization and debridement, pECM powder was placed over the wound bed at the patient’s bedside followed by petroleum gauze (Figure 2). Twice daily dressing changes were performed utilizing a solution of hypochlorous acid (Vashe, Urgo Medical) as a moistening agent followed by dry gauze.
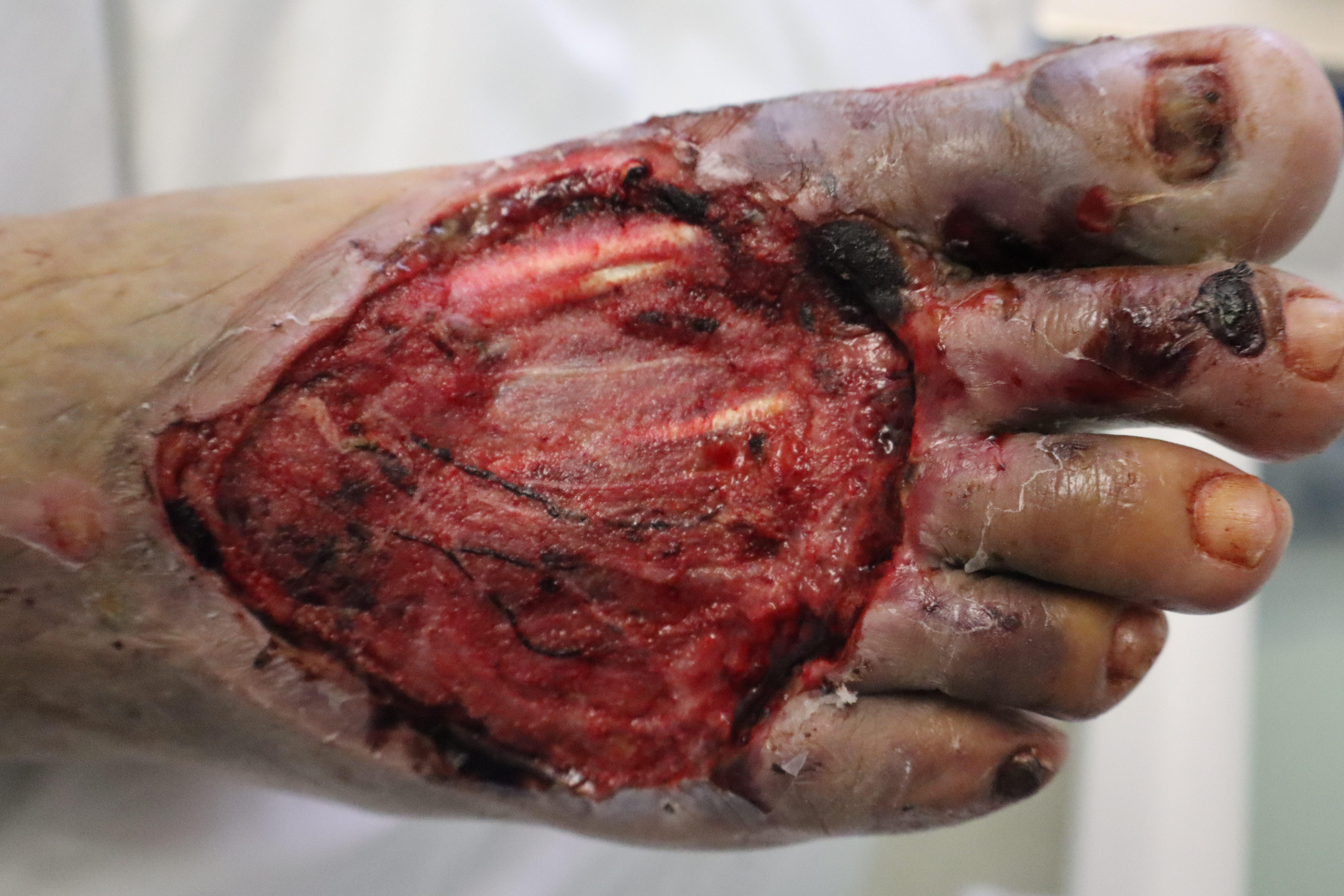
Figure 1. Initial dorsal foot wound after operative debridement.
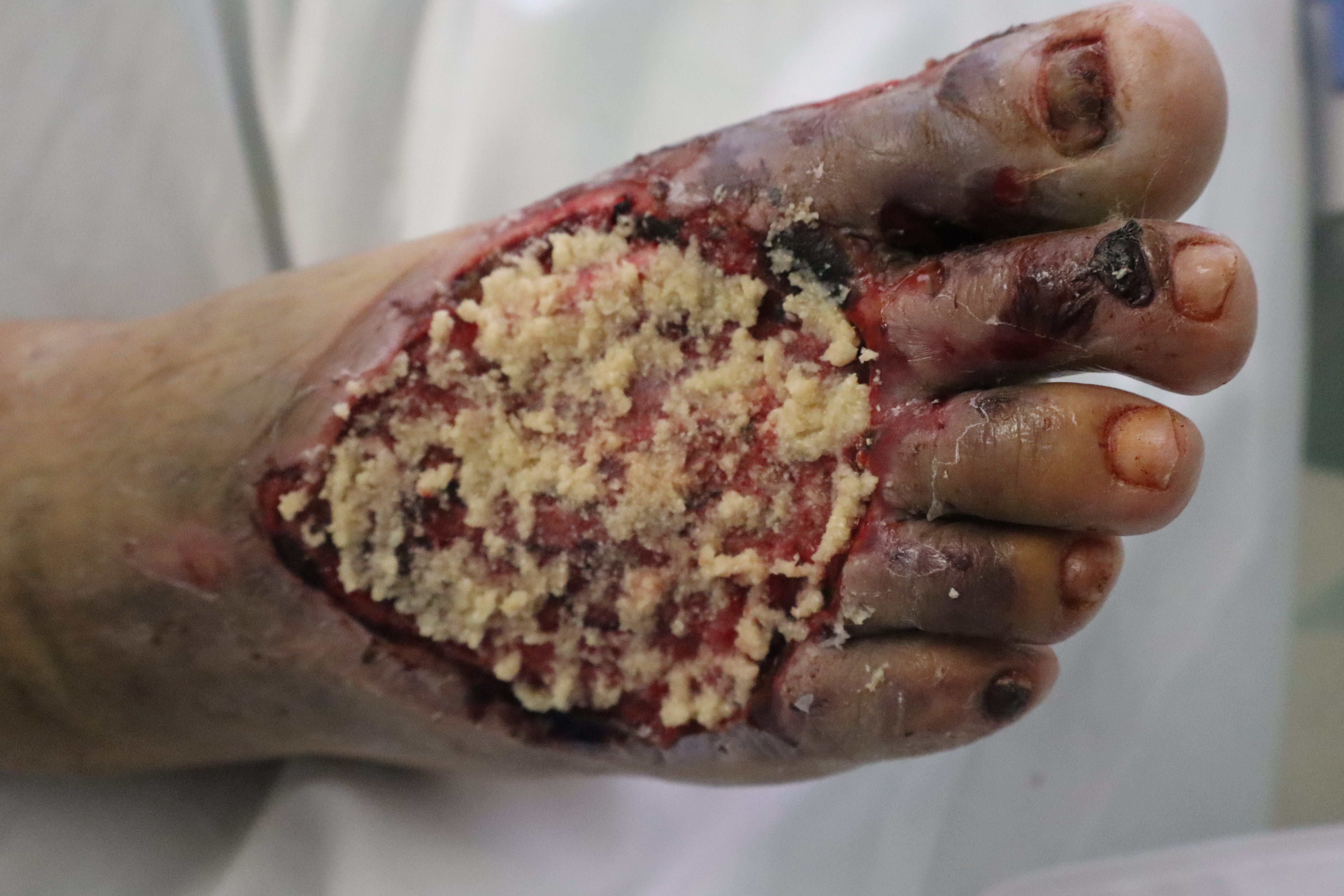
Figure 2. Application of porcine-derived extracellular matrix powder.
The matrix was well tolerated, and the wound was closely observed over the following days. By POD 11, granulation tissue had fully matured, covering the wound bed with uniform vascularity and appropriate exudate levels. Based on this assessment, a STSG was placed (Figure 3). The graft site was monitored for signs of hematoma, seroma, or infection, and none were observed. Early signs of graft integration, including capillary refill and adherence to the wound bed, were evident by POD 3.
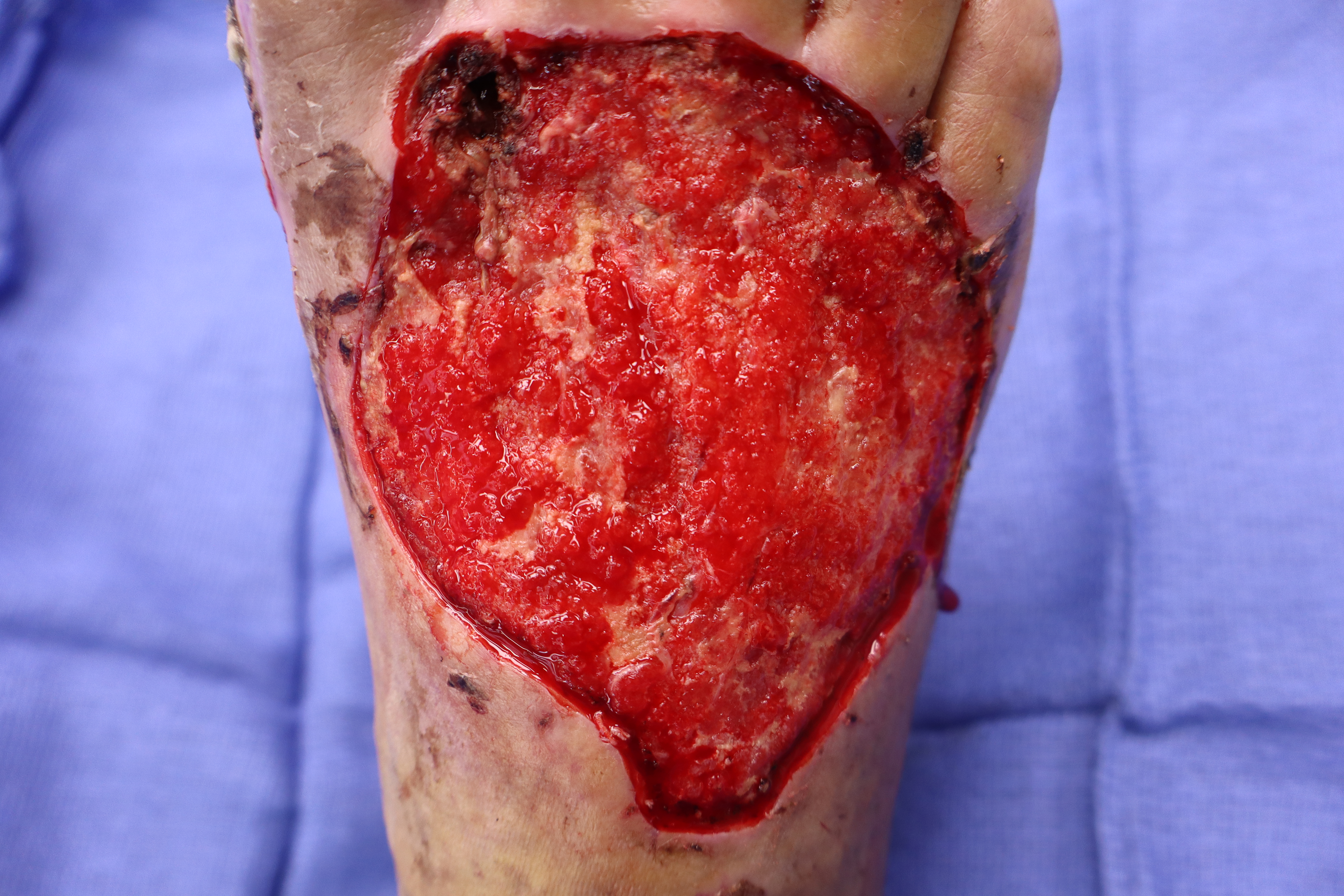
Figure 3. Robust granulation tissue present over the wound bed at post-application day 11.
The patient experienced no postoperative complications such as graft sloughing, necrosis, or cellulitis. Pain at the donor and recipient sites was managed effectively with standard analgesics. Over the next several days, the dressing regimen supported ongoing healing, and at discharge (POD 5) the graft demonstrated excellent adherence with intact borders and minimal drainage.
Follow-up assessments revealed continued success. At 3 months postoperatively, the graft had matured without contracture or significant hypertrophic scarring. The cosmetic appearance was favorable, and the patient regained stable ambulatory function. There was no need for revision grafting, and the patient reported satisfaction with the final outcome (Figure 4).
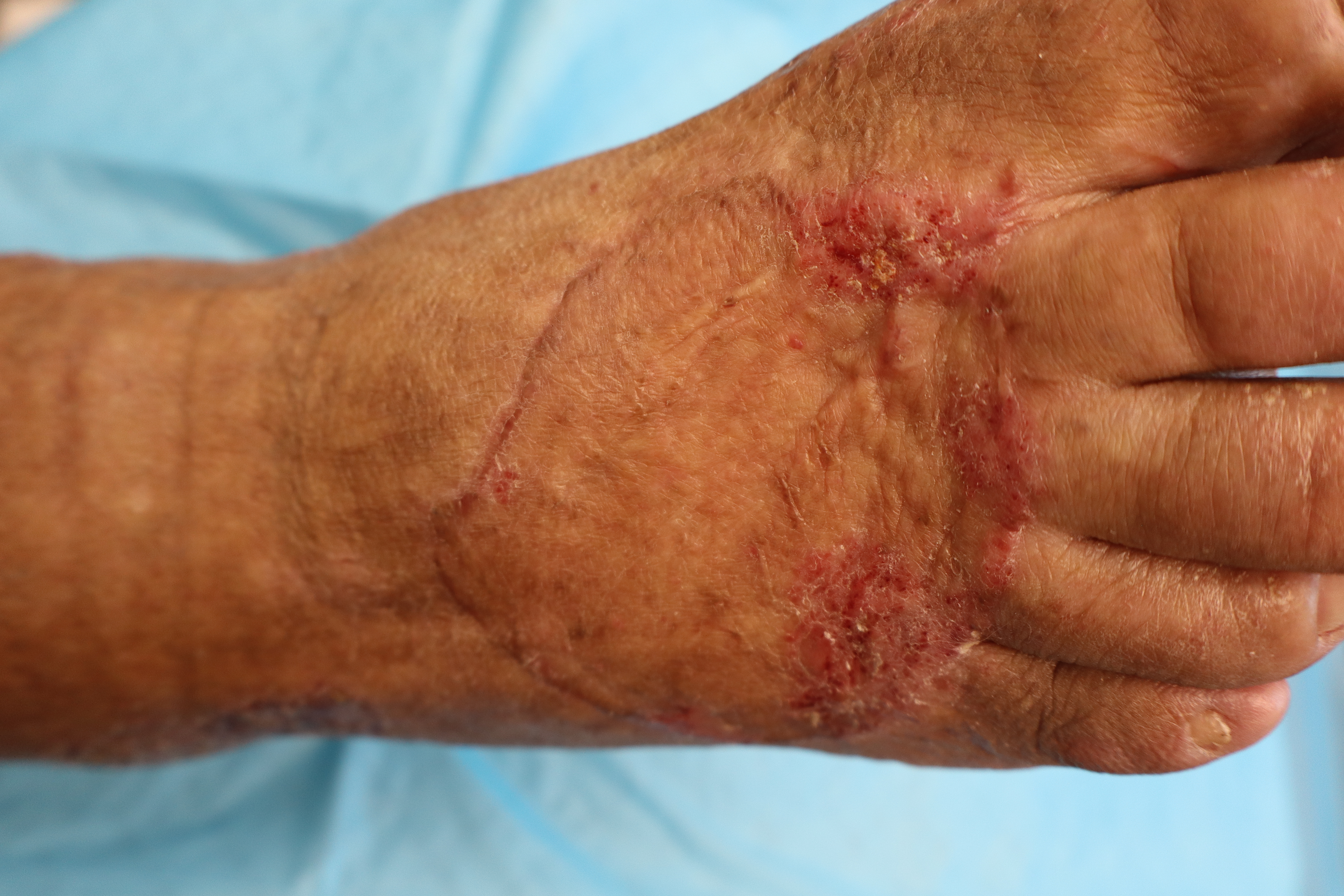
Figure 4. Dorsal foot 3 months postoperative split-thickness skin graft.
Case 2: extensive thermal burn injury
A 52-year-old female with a history of thermal injury underwent excision of function-limiting neck scar contracture with reconstruction with a free latissimus dorsi myocutaneous flap (Figure 5). Given the complexity of the wound surface and the importance of prompt vascularized tissue formation, pECM powder was applied intraoperatively to the flap bed to promote early granulation and enhance graft readiness. Figure 6 depicts intraoperative placement of the pECM over the latissimus flap following contracture release. This was undertaken prior to grafting in order to enhance vascularized granulation of the flap surface (Figure 6).
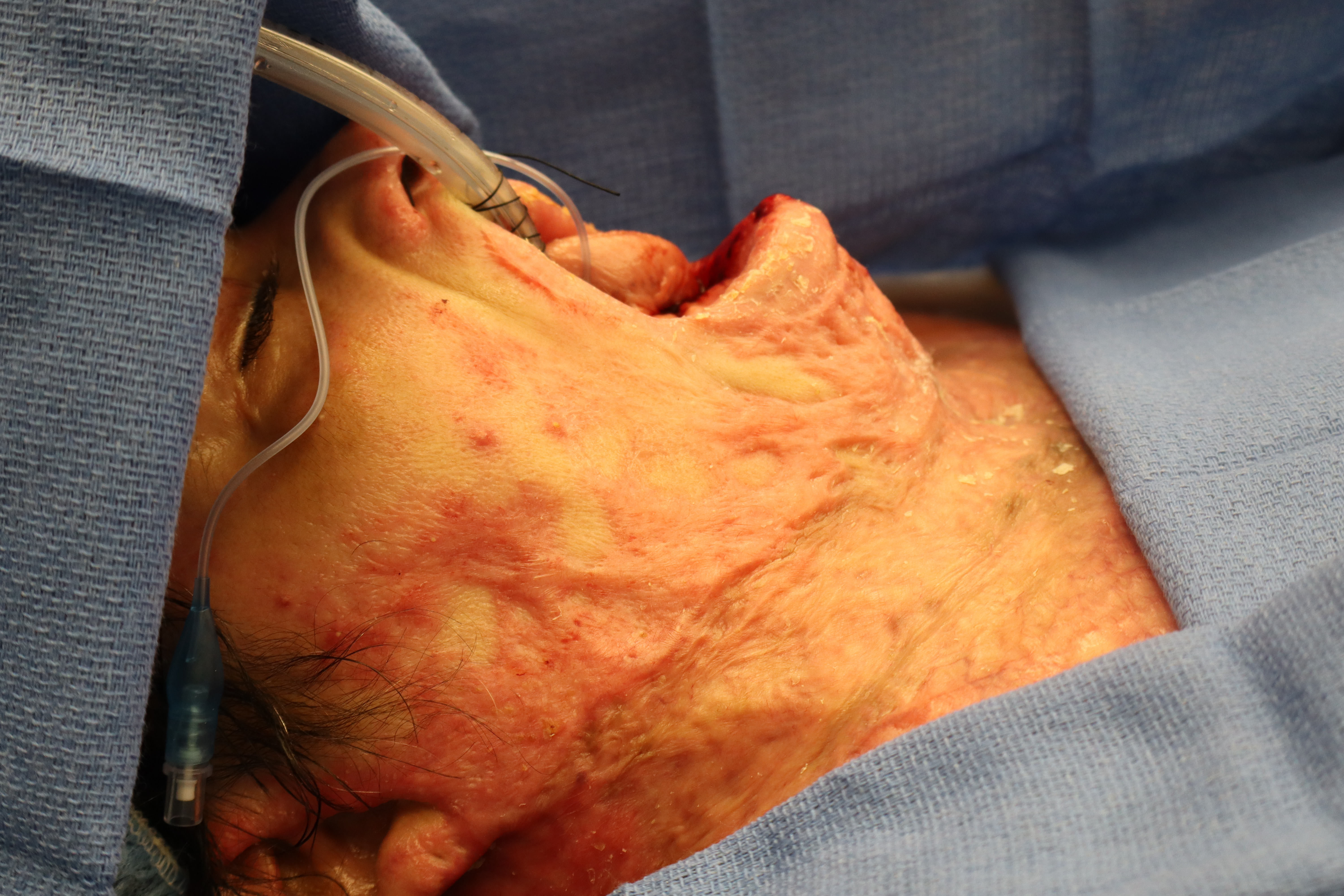
Figure 5. Severe neck burn contracture prior to excision with reconstruction.
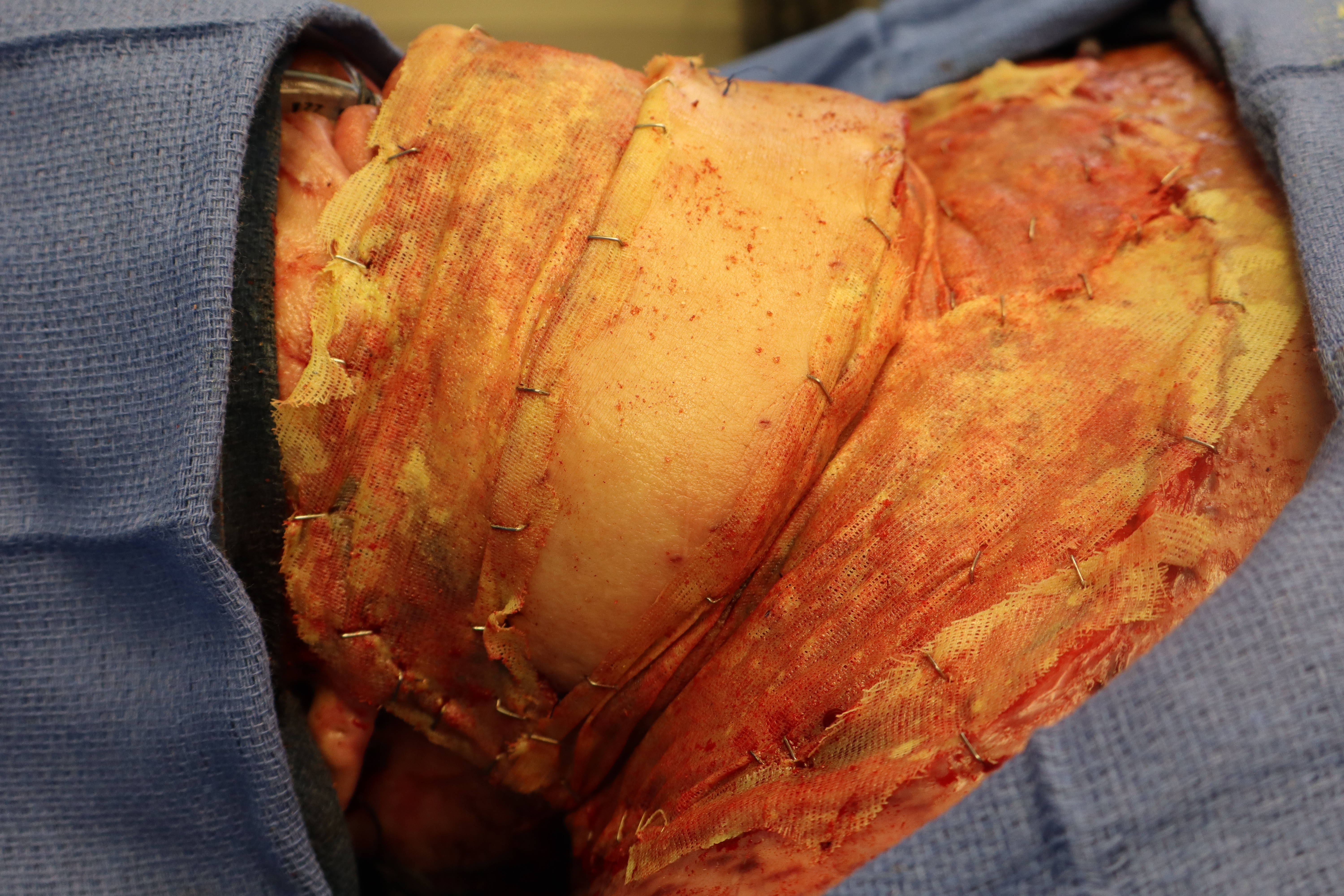
Figure 6. Reconstruction of neck defect with latissimus dorsi myocutaneous flap with application of porcine-derived extracellular matrix powder and gauze dressings.
Daily wound evaluation demonstrated progressive matrix integration without excessive exudate, foul odor, or signs of infection. By day 9 post-application, the site displayed a healthy granulation profile suitable for grafting. A meshed STSG along with epidermal autograft was applied over the wound bed. Postoperative recovery was notable for full graft adherence, with no evidence of seroma, partial take, or edge necrosis. The patient remained in the hospital under multidisciplinary burn and reconstructive care for additional optimization of graft donor sites and to initiate rehabilitation.
Two weeks after discharge, outpatient follow-up confirmed complete graft survival and durable integration with the underlying flap. The reconstructed region showed stable coverage, pliability, and preservation of neck mobility. No complications such as wound dehiscence, flap failure, or infection were encountered. At 3-month follow-up, the patient showed appropriate graft take with favorable contour, pigmentation, and improved range of motion, contributing to a meaningful enhancement in quality of life and cosmetic satisfaction (Figure 7).
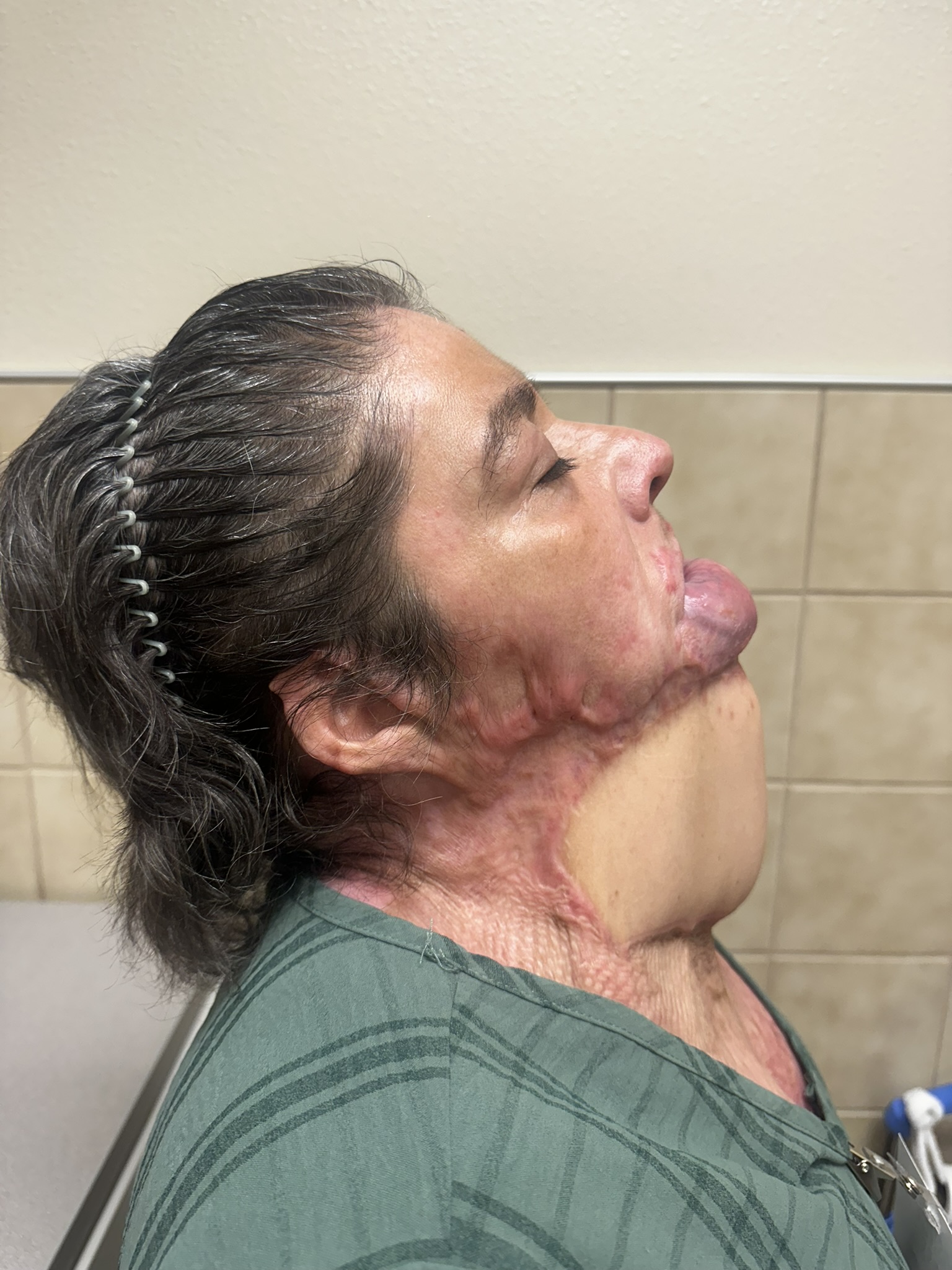
Figure 7. Surgical site 3 months postoperative showing skin graft adherence with improved range of motion.
Case 3: free omental flap reconstruction of extensive upper extremity defect
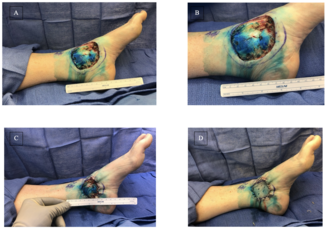
A 46-year-old male with history of right upper extremity crush injury with degloving underwent soft tissue reconstruction for extensive defect using a free omental flap (Figures 8 and 9). After microvascular anastomosis and flap inset, pECM powder was applied directly on top of the flap followed by Xeroform gauze followed by a negative pressure wound therapy device utilizing a white sponge set at -75 mm Hg (Figure 10). Bedside negative pressure therapy change was performed starting POD 5 and then every 3 days until the decision was made to perform skin grafting on POD 13 (Figure 11). Histological section revealed neovascularization and collagen deposition within a fibrin network (Figure 12). A 0.016-inch STSG was placed on the flap, followed by moist dressings. The patient had an uneventful recovery and achieved complete healing at 3 months (Figure 13).
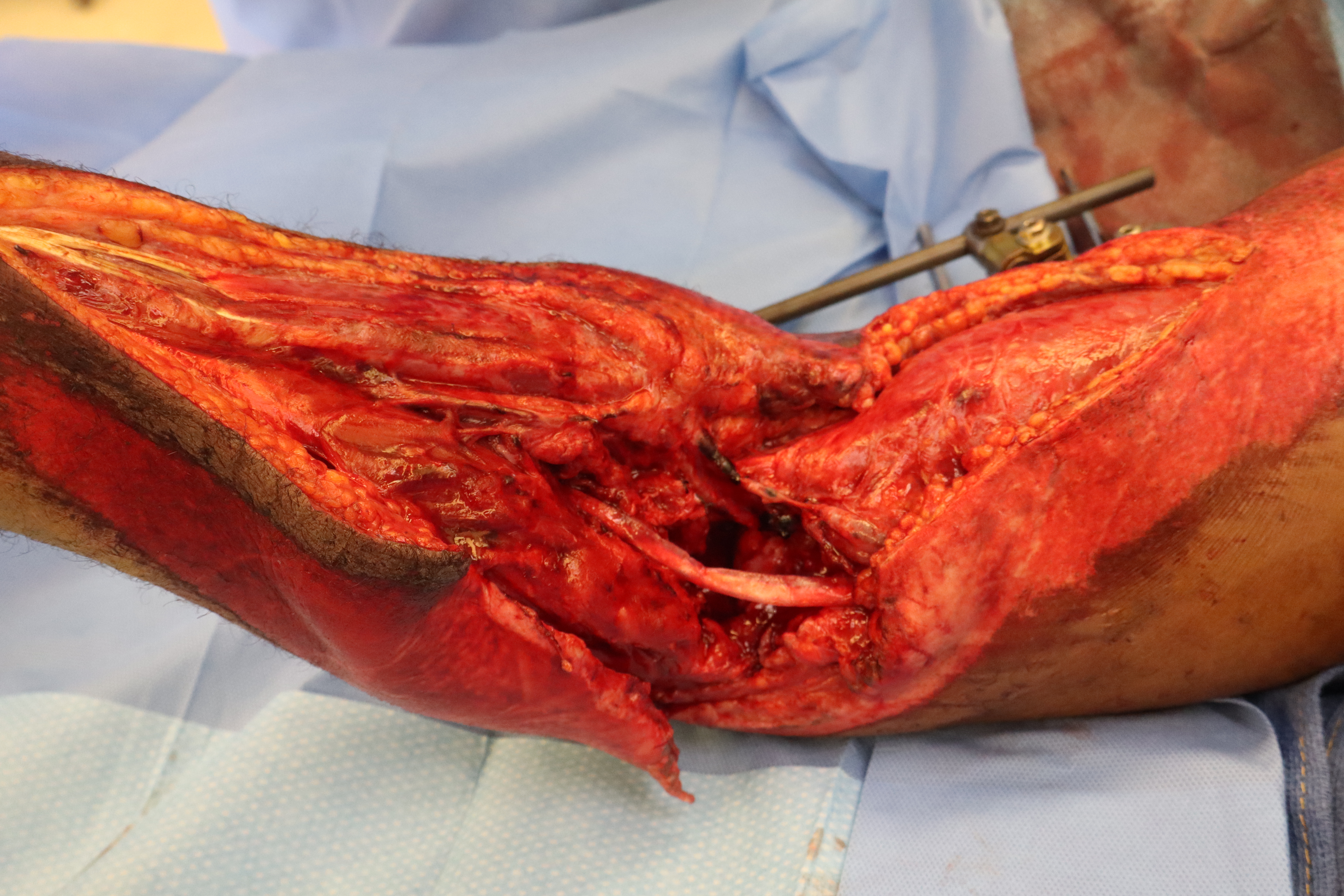
Figure 8. Initial right upper extremity traumatic defect with exposed radial nerve and radial artery.
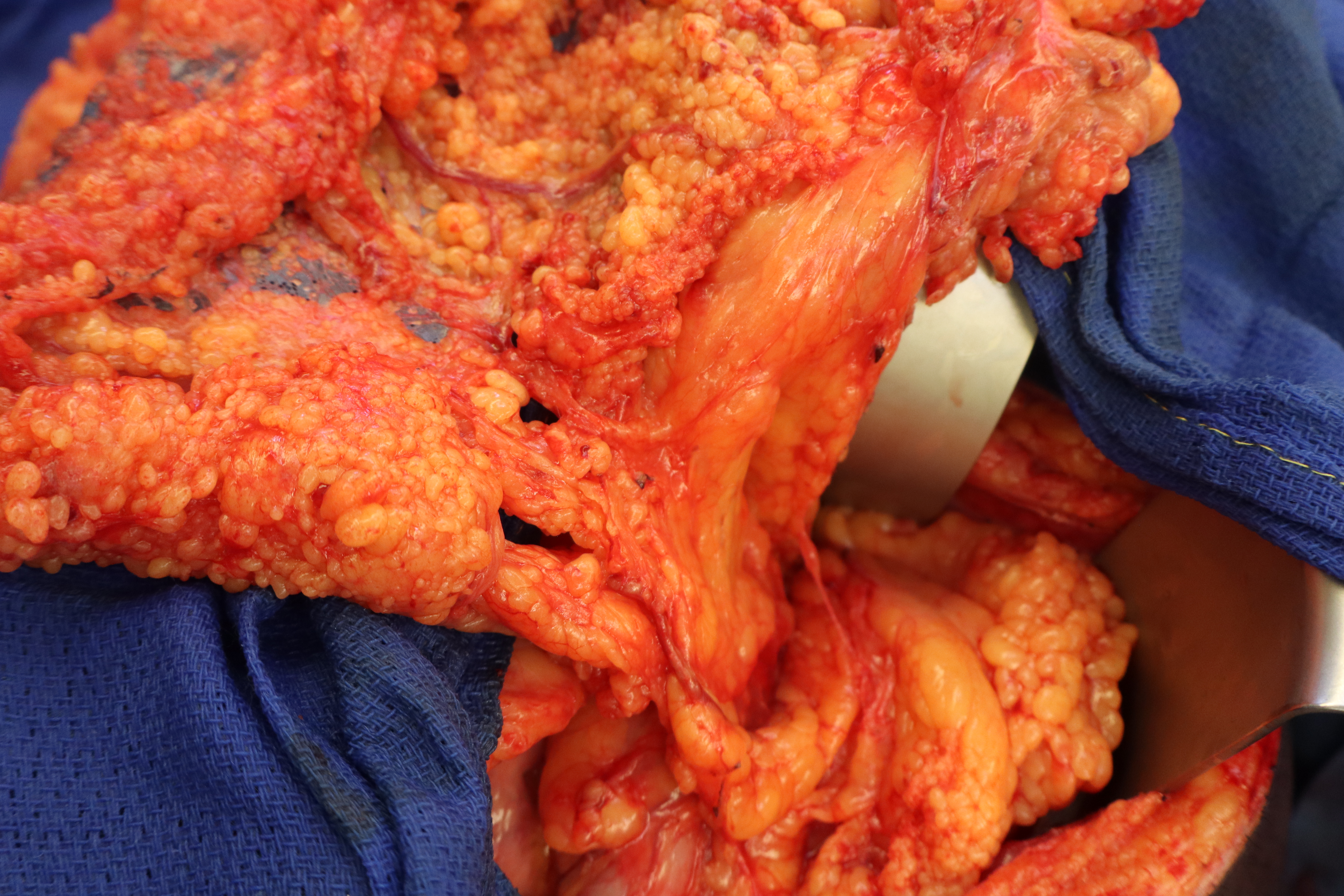
Figure 9. Omental flap harvest.
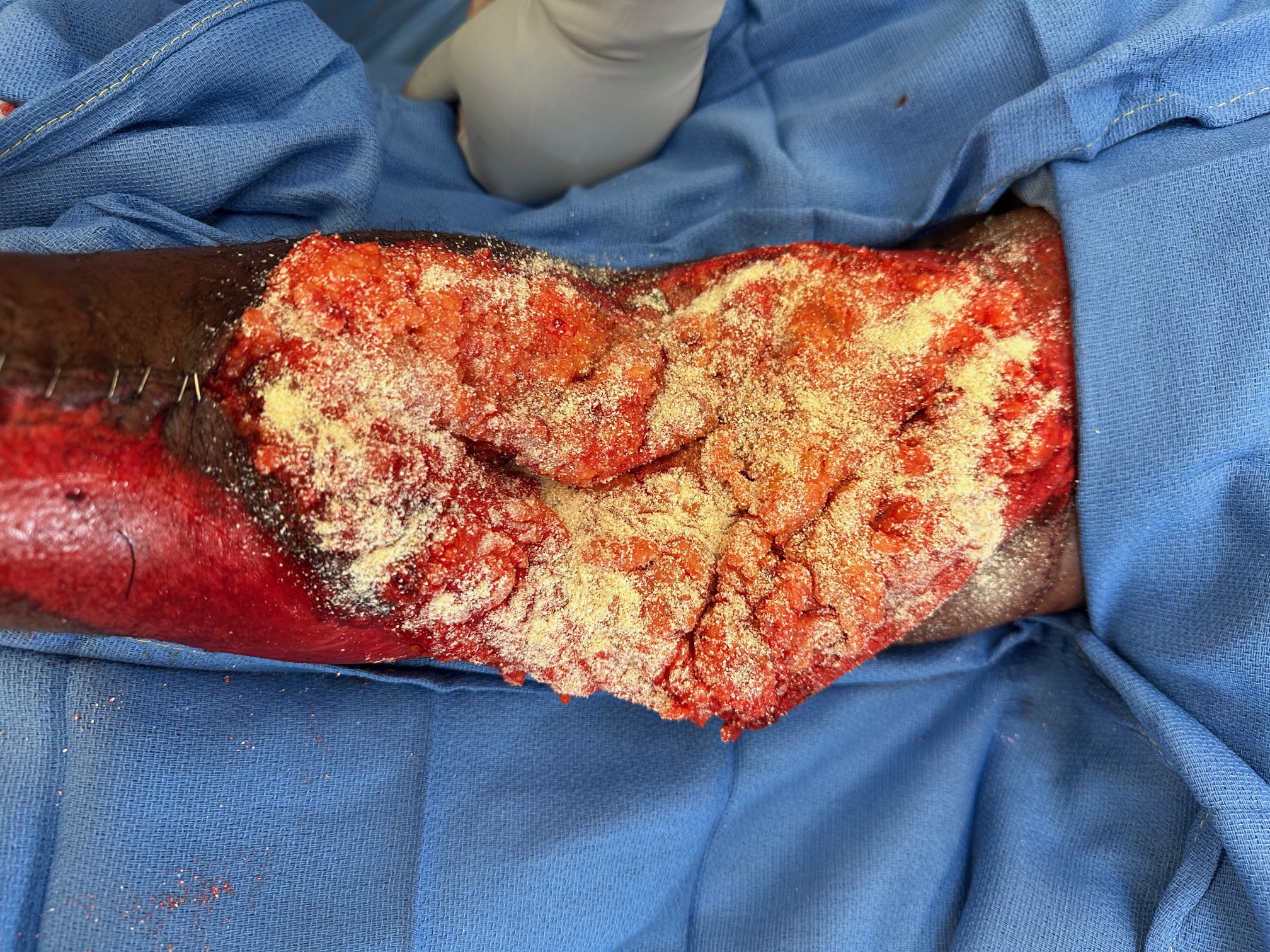
Figure 10. Application of porcine-derived extracellular matrix powder to free omental flap.
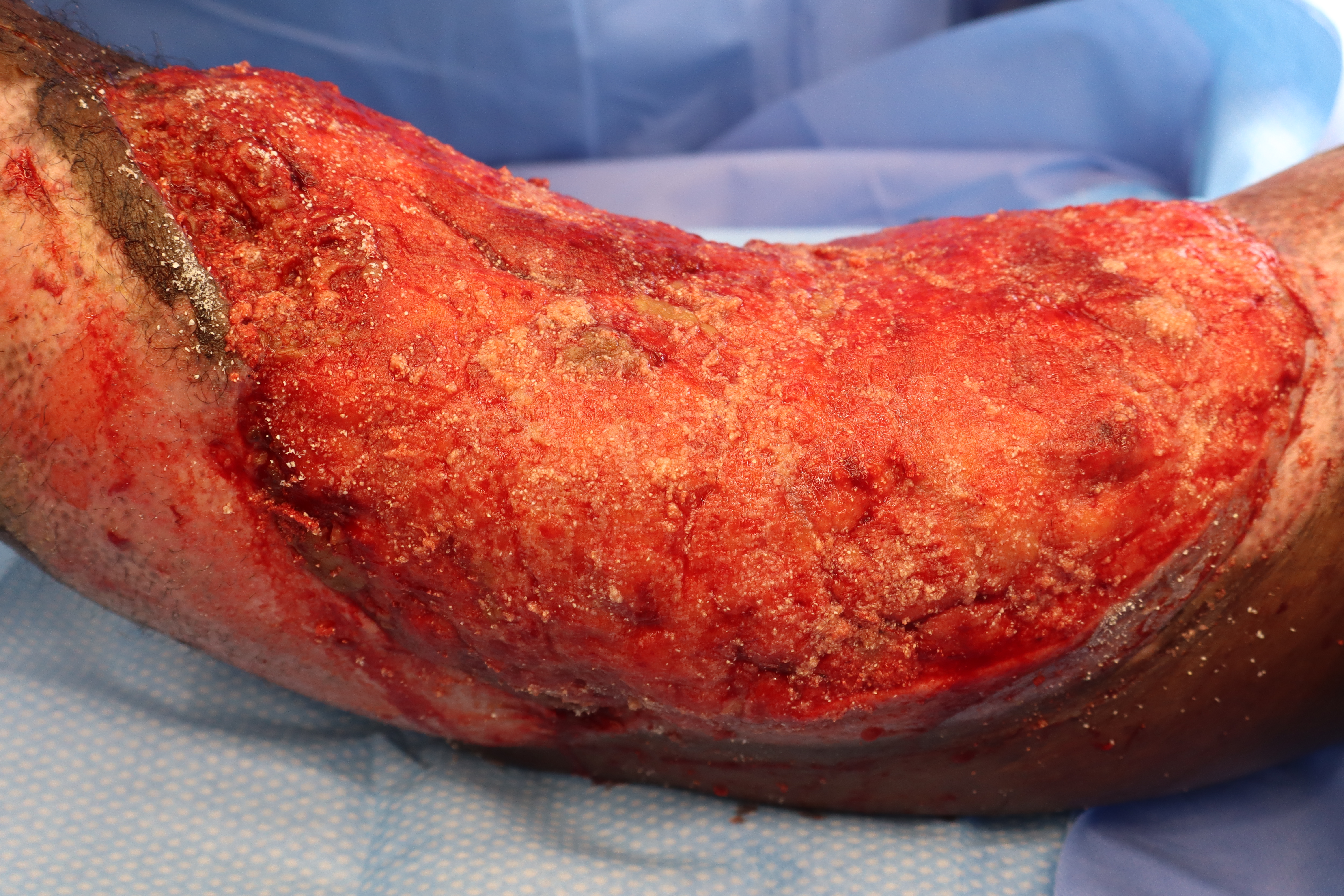
Figure 11. Formation of granulation tissue over free omental flap on postoperative day 13.
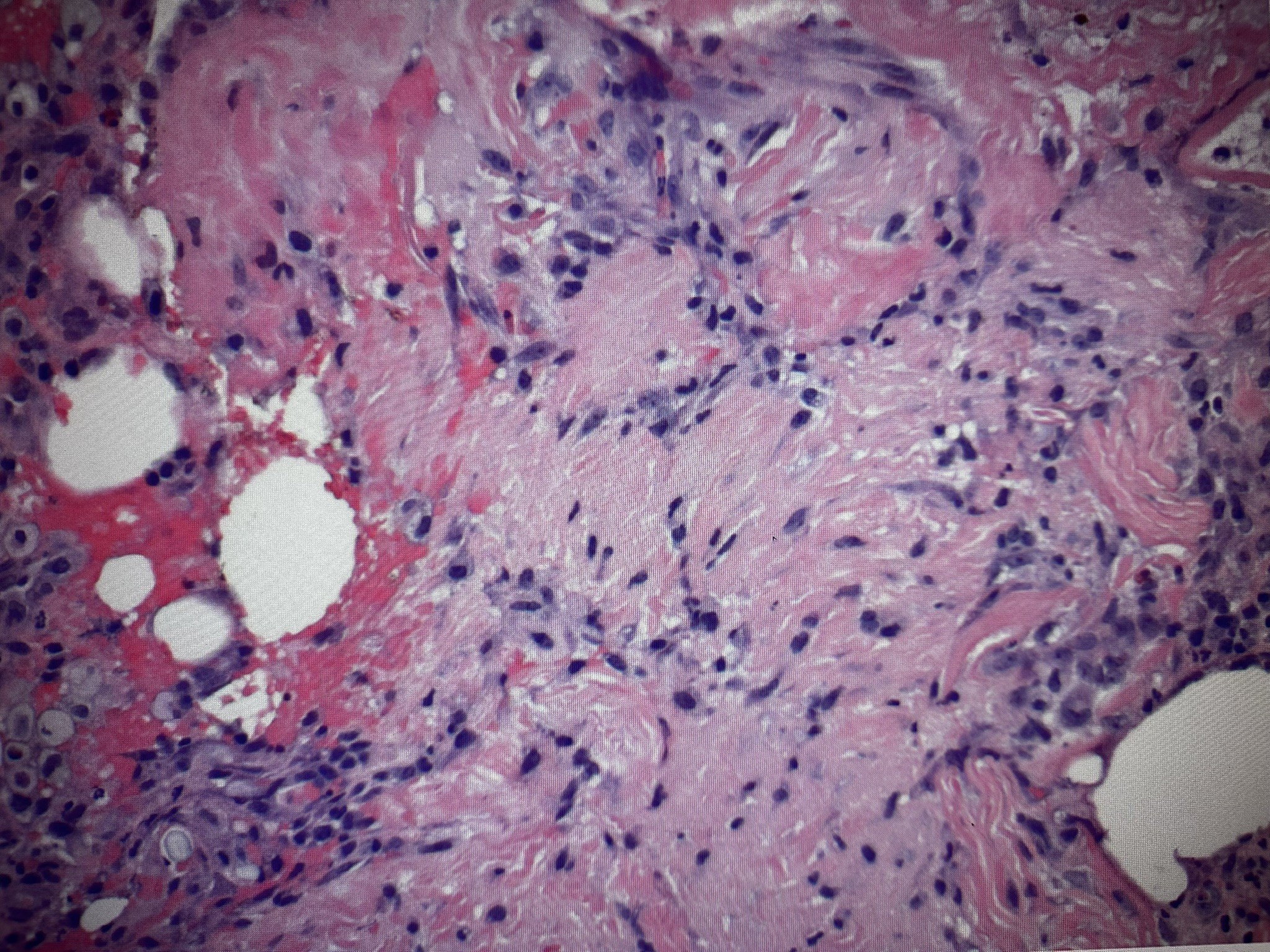
Figure 12. Histological sectioning of granulation tissue illustrating neovascularization and collagen deposition within a fibrin network.
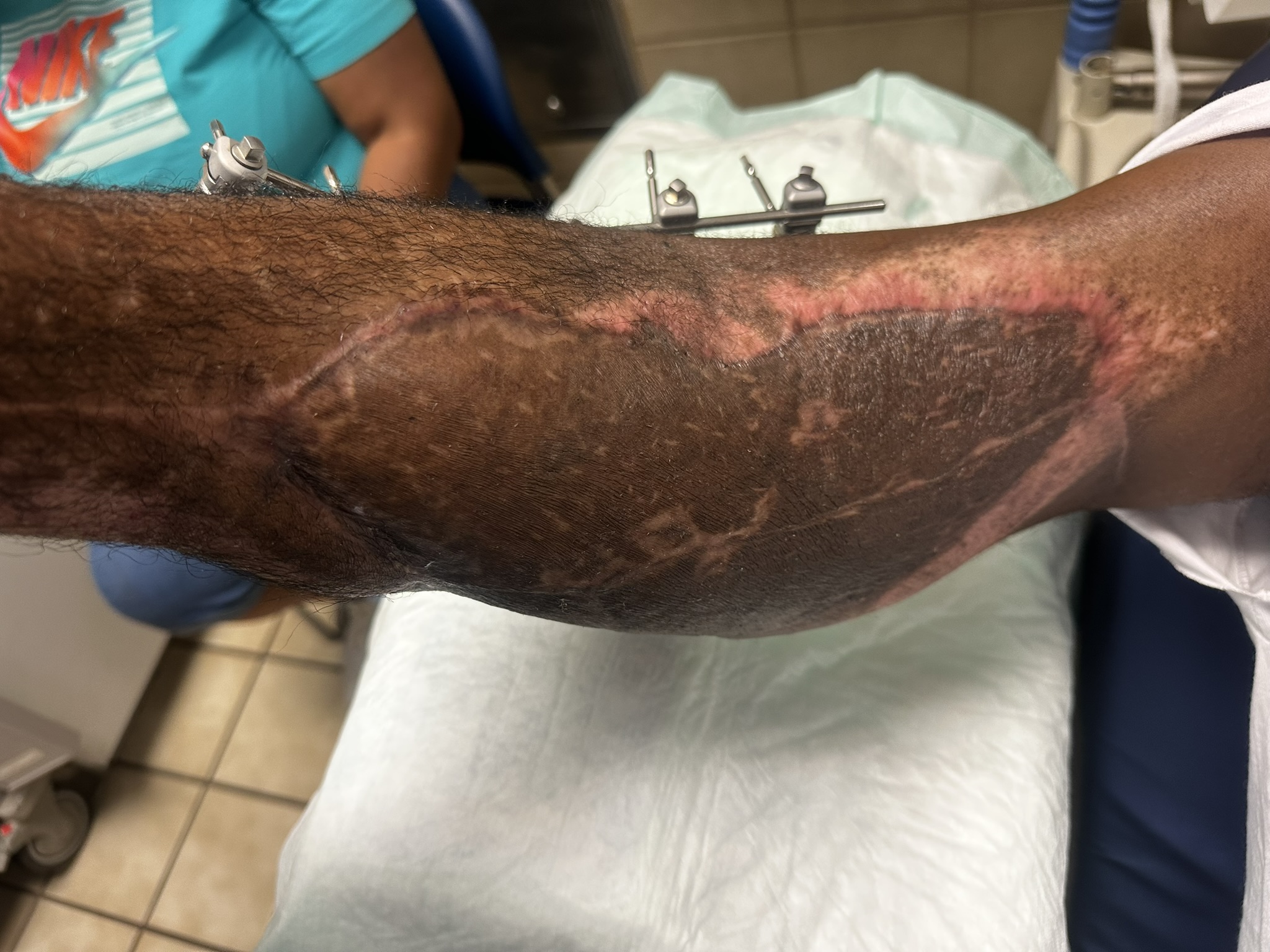
Figure 13. Surgical site 3 months postoperatively illustrating robust adherence of skin graft.
Discussion
The restoration of durable, vascularized soft tissue in complex wounds remains a cornerstone of reconstructive surgery. Traditional dermal substitutes such as BWM and BTM have long served this purpose; however, the extended time to granulation tissue maturity with these options may delay definitive skin grafting and increase patient morbidity.7-8 This study elucidates the utilization of pECM powder as an alternative approach to expedite wound bed preparation. Our findings in this series suggests that the usage of pECM powder fosters a rapid regenerative response, enabling skin grafting success within a significantly shorter window compared with conventional products.
Acceleration of granulation tissue formation
One of the most critical advantages observed with pECM powder was the acceleration of granulation tissue formation. In all cases reported, a robust wound bed was achieved within 9 to 13 days, which is markedly faster than the average timeframes noted in previous literature both for BTM and BWM.7-9 This aligns with previous literature regarding the efficacy of ECM products in granulation tissue formation. A study by Anikumar et al showed that an injectable porcine cholecyst-derived ECM hydrogel significantly accelerated wound healing in animal models, promoting a favorable immune response characterized by M2 macrophage dominance and collagen remodeling, both of which are vital for early granulation tissue formation.15 Additionally, Wang et al outlined improved wound healing outcomes in severely damaged extremities using a porcine acellular dermal matrix, which provided a bioactive scaffold capable of stimulating angiogenesis and epithelialization.16
Comparison with other ECM-based strategies
ECMs have emerged as a key player in regenerative medicine, with porcine urinary bladder matrix (UBM) and cholecyst-derived scaffolds among the most extensively studied. One study reported complete epithelialization of chronic wounds using UBM in a small patient cohort, with limited scar tissue and a high rate of wound closure.17 Similarly, in a comparative animal study, it was identified that porcine cholecyst ECM induced more effective healing than conventional bovine collagen sheets, with superior wound contraction and granulation quality.18 These results, along with our current study, emphasize that ECM origin and structure significantly affect healing outcomes. Porcine matrices appear to establish an optimal balance between structural integrity, host integration, and bioactivity—thus promoting their continual investigation as an ideal dermal substitute.
Clinical implications
The ability to reduce the time to grafting has significant implications for patient outcomes and health care efficiency. Faster wound closure minimizes infection risk, reduces pain, and allows for deceased length of hospitalization.19-21 This is especially critical in vulnerable populations such as elderly or immunocompromised patients, evidenced by the 80-year-old male in case 1, where prolonged wound exposure could have led to severe systemic complications. In burn wound care, as illustrated in case 2, early intervention with biologically active matrices is able to mitigate long-term morbidity and reduce need for repeated surgical debridements. Additionally, the format of pECM powder simplifies its storage, handling, and intraoperative use compared to multilayered synthetic matrices, which often require shaping and suturing. This ease of use may broaden its utility in various surgical settings, including outpatient wound clinics and resource-limited environments.
Consideration of immediate autografting
In some situations, immediate split-thickness autografting following debridement or flap inset may be feasible. However, in the cases presented here, immediate grafting was deemed less optimal due to wound complexity, irregular wound topography, and the need for enhanced vascularization of the surface. For example, in case 2, a direct autograft over the latissimus flap could have risked partial loss due to insufficient early vascularization. The application of pECM powder provided a scaffold that rapidly vascularized and in turn supported complete graft take.
Host response and risk of rejection
The brisk vascularized granulation observed in our cases does not represent rejection of the pECM utilized but, instead, a constructive remodeling response. Prior work on porcine-derived matrices has demonstrated that the ECM is resorbed and replaced with host tissue through a primarily proregenerative immune response.15–16 No clinical evidence of rejection, excessive inflammation, or graft failure was observed in these patients.
Cost considerations
The relative expense of intervention is another important factor in matrix selection. Dermal regeneration templates can be costly, both in terms of material price and prolonged hospital stay required prior to grafting. The pECM powder is generally less expensive per application as a powdered matrix and requires minimal storage or preparation compared to bilayer constructs. Furthermore, the accelerated readiness for grafting suggests the potential for both material and hospitalization cost savings. Additional formal economic comparisons are warranted in future studies to assess these differences more thoroughly.
Limitations
While the results from this case series are promising regarding the utility of pECM powder to improve clinical outcomes of wound patients, they are inherently limited by the small sample size and lack of a control group. Larger, randomized controlled trials are needed to quantify the efficacy, safety, and cost-effectiveness of pECM powder in comparison to established wound care products to better characterize the benefits. Future studies should also investigate histologic changes, immunologic response, and long-term cosmetic outcomes to holistically assess the efficacy of pECM powder. There is also potential to explore combination therapies, such as pairing ECM matrices with autologous stem cells or growth factors, which may provide further elucidation as to which wound healing strategies may yield the best patient outcomes. Finally, future investigations should compare direct immediate autografting versus matrix-assisted granulation in order to elucidate which wound types and intermediary scaffold can provide measurable benefit.
Conclusions
This case series demonstrates that pECM powder can serve as an effective dermal template, accelerating granulation tissue formation and enabling earlier split-thickness skin grafting in patients with complex wounds. Compared to literature on conventional dermal substitutes such as BWM and BTM, pECM powder significantly reduced the time to wound closure, achieving definitive grafting readiness within 9 to 13 days.
These findings underscore the potential role of acellular xenograft matrices in both acute and subacute wound management, especially in patients with limited reconstructive options or heightened surgical risk. Although promising, further large-scale, prospective studies are necessary to establish definitive efficacy and explore long-term outcomes in diverse wound patient populations.
Acknowledgments
Authors: Henry Krasner, BS1*; Joshua Khorsandi, BS1*; Dustin Dillon, MD2; Noama Iftekhar, MD2; Joshua MacDavid, MD3
Affiliations: 1Kirk Kerkorian School of Medicine at the University of Nevada, Las Vegas, Las Vegas, Nevada; 2Department of Surgery, Kirk Kerkorian School of Medicine at the University of Nevada, Las Vegas, Las Vegas, Nevada; 3Department of Plastic Surgery, Kirk Kerkorian School of Medicine at the University of Nevada, Las Vegas, Las Vegas, Nevada
*Joint first coauthorship
Correspondence: Henry Krasner; krasnh1@unlv.nevada.edu
Ethics: Patient consent was confirmed for the inclusion in this study and the utilization of images.
Disclosures: The authors disclose no relevant financial or nonfinancial interests.
References
- Alhajj M, Goyal A. Physiology, granulation tissue. In: StatPearls. StatPearls Publishing; Updated October 24, 2022. Accessed March 11, 2026. https://www.ncbi.nlm.nih.gov/books/NBK554402/
- Lagus H, Kankuri E, Nuutila K, Juteau S, Sarlomo-Rikala M, Vuola J. Induced granulation tissue but not artificial dermis enhances early host-graft interactions in full-thickness burn wounds. World J Surg. 2018;42(4):981-991. doi:10.1007/s00268-017-4270-6
- Rudolph R. Inhibition of myofibroblasts by skin grafts. Plast Reconstr Surg. 1979;63(4):473-480. doi:10.1097/00006534-197904000-00005
- Takabayashi Y, Ishihara M, Sumi Y, Takikawa M, Nakamura S, Kiyosawa T. Platelet-rich plasma-containing fragmin-protamine micro-nanoparticles promote epithelialization and angiogenesis in split-thickness skin-graft donor sites. J Surg Res. 2015;193(1):483-491. doi:10.1016/j.jss.2014.08.011
- Teh BT. Why do skin grafts fail? Plast Reconstr Surg. 1979;63(3):323-332. doi:10.1097/00006534-197903000-00005
- Hierner R, Degreef H, Vranckx JJ, Garmyn M, Massagé P, Van Brussel M. Skin grafting and wound healing—the dermato-plastic team approach. Clin Dermatol. 2005;23(4):343-352. doi:10.1016/j.clindermatol.2004.07.028
- Grande PK, Hill D, McElfresh J, Velamuri R, Liu X. Systematic review and meta-analysis of biodegradable temporizing matrix application for complex wound reconstruction. J Burn Care Res. 2025;46(1):82-89. doi:10.1093/jbcr/irae081
- Kidd T, Kolaityte V, Bajaj K, Wallace D, Izadi D, Bechar J. The use of NovoSorb biodegradable temporising matrix in wound management: a literature review and case series. J Wound Care. 2023;32(8):470-478. doi:10.12968/jowc.2023.32.8.470
- Helgeson MD, Potter BK, Evans KN, Shawen SB. Bioartificial dermal substitute: a preliminary report on its use for the management of complex combat-related soft-tissue wounds. J Orthop Trauma. 2007;21(6):394-399. doi:10.1097/BOT.0b013e318070c028
- Farabi B, Roster K, Hirani R, Tepper K, Atak MF, Safai B. The efficacy of stem cells in wound healing: a systematic review. Int J Mol Sci. 2024;25(5):3006. doi:10.3390/ijms25053006
- Duscher D, Barrera J, Wong VW, et al. Stem cells in wound healing: the future of regenerative medicine? A mini-review. Gerontology. 2016;62(2):216-225. doi:10.1159/000381877
- Zhao Y, Wang M, Liang F, Li J. Recent strategies for enhancing the therapeutic efficacy of stem cells in wound healing. Stem Cell Res Ther. 2021;12(1):588. doi:10.1186/s13287-021-02657-3
- Tartarini D, Mele E. Adult stem-cell therapies for wound healing: biomaterials and computational models. Front Bioeng Biotechnol. 2016;3:206. doi:10.3389/fbioe.2015.00206
- Chen J, Han C, Zhang L. Zhonghua Zheng Xing Wai Ke Za Zhi. 2002;18(5):271-272.
- Pratheesh KV, Nair RS, Purnima C, et al. An injectable hydrogel of porcine cholecyst extracellular matrix for accelerated wound healing. J Biomed Mater Res A. 2025;113(1):e37795. doi:10.1002/jbm.a.37795
- Zhang Z, Lv L, Mamat M, Chen Z, Liu L, Wang Z. Xenogenic (porcine) acellular dermal matrix is useful for the wound healing of severely damaged extremities. Exp Ther Med. 2014;7(3):621-624. doi:10.3892/etm.2014.1490
- Kimmel H, Rahn M, Gilbert TW. The clinical effectiveness in wound healing with extracellular matrix derived from porcine urinary bladder: a case series on severe chronic wounds. J Am Coll Certif Wound Spec. 2010;2(3):55-59. doi:10.1016/j.jcws.2010.11.002
- Shakya P, Sharma AK, Kumar N, et al. Bubaline cholecyst-derived extracellular matrix for reconstruction of full-thickness skin wounds in rats. Scientifica (Cairo). 2016;2016:2638371. doi:10.1155/2016/2638371
- Quinn RH, Wedmore I, Johnson E, et al. Wilderness Medical Society practice guidelines for basic wound management in the austere environment. Wilderness Environ Med. 2014;25(3):295-310. doi:10.1016/j.wem.2014.04.005
- Forsch RT, Little SH, Williams C. Laceration repair: a practical approach. Am Fam Physician. 2017;95(10):628-636.
- Singh D, Chopra K, Sabino J, Brown E. Practical things you should know about wound healing and vacuum-assisted closure management. Plast Reconstr Surg. 2020;145(4):839e-854e. doi:10.1097/PRS.0000000000006652