Rethinking Compression: Expanding Decongestive Therapy With Innovative Garment Solutions
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of Wounds or HMP Global, their employees, and affiliates.
Abstract
Background. Access to skilled lymphedema care is increasingly limited due to workforce shortages, financial constraints, and logistical barriers. Although compression bandaging remains the standard for the intensive phase and compression garments for maintenance, their complexity and reliance on trained clinicians pose challenges for many patients. This article explores the use of an elastic nighttime garment (ENG) and an adjustable wrap device (AWD) in both phases to optimize outcomes. The ENG used in this analysis is a specific low‑profile, elastic, lymphatic alternating pressure profile–based design. The AWD used is constructed using overlapping, knitted, short-stretch bands rather than laminate textile. Objective. To evaluate the clinical viability of a dual-layer, patient-managed compression system that combines an ENG with an AWD as an alternative to traditional bandaging during the intensive phase of complete decongestive therapy (CDT) and as a tool for long-term maintenance. Methods. This prospective case-based analysis draws from 5 representative patients with complex lymphedema presentations and barriers to conventional CDT. Each case details diagnosis, intervention protocol, and outcomes using the dual-layer system. Compression combinations were tailored to individual needs, with emphasis on self-management, mobility, and wound care compatibility. Results. All patients achieved measurable limb volume reduction, improved adherence, and enhanced independence. The system facilitated transition to custom garments, reduced clinic visit frequency, and supported wound healing and mobility in diverse clinical scenarios. Conclusion. The dual-layer ENG plus AWD system offers a scalable, evidence-informed alternative to traditional compression bandaging. It enhances access, supports patient autonomy, and bridges intensive therapy with sustainable self-management, which is particularly valuable in underserved and resource-limited settings.
Compression therapy remains the cornerstone of lymphedema management, during both the intensive and maintenance phases of complete decongestive therapy (CDT).1-5 Typically, the intensive phase relies on skilled application of multilayer, multicomponent bandaging systems, often involving 4 to 12 short-stretch bandages layered over foam padding and a hygienic interface, and multiple treatment visits during the week.4,5 This compression application is clinically effective, offering high working pressure and low resting pressure to stimulate lymphatic flow and reduce limb volume.4,5 Moreover, it can be customized to incorporate specialized textiles, such as foam inserts, chip pads, or ridged materials, that create an alternating pressure profile.3,5 The lymphatic alternating pressure profile (LAPP) has been cited in clinical literature for its effect on enhancing lymphatic contractility, stimulating dermal lymphatic channels, and positively influencing fibrotic tissue by softening and improving tissue texture.6-8 These effects support improved tissue pliability and restoration of integumentary function. However, traditional bandaging techniques remain labor-intensive, requiring skilled clinicians for precise application and frequent reapplication as limb volume fluctuates, contributing to increased treatment costs and financial burden for patients, particularly those with breast cancer–related lymphedema.12 In patients with chronic wounds and lower extremity lymphedema, compression bandaging often necessitates frequent clinic visits for reapplication and monitoring.13 These repeated visits add to the clinical workload and impose significant logistical and financial strain on patients.12,13
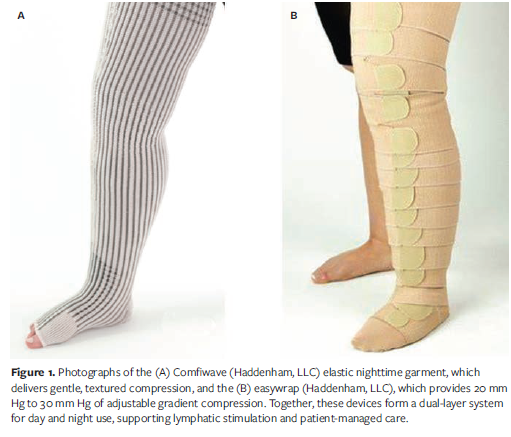
While bandaging is the standard for the intensive phase, compression garments are essential for long-term maintenance, ensuring sustained limb volume control and patient independence.2,14,15 Both compression bandages and compression garments are now covered under the Lymphedema Treatment Act, a federal law that took effect in 2024 to ensure Medicare coverage for compression devices prescribed to treat lymphedema.15 The passage of the Lymphedema Treatment Act represents a landmark step in expanding coverage for compression bandages and garments, affirming their role as medically necessary interventions. Despite this policy advance, however, access to this gold standard approach remains increasingly constrained. A shortage of trained clinicians, coupled with financial barriers such as copays and limited insurance coverage as well as geographical and logistical barriers to frequent clinic visits, leaves many patients unable to receive the frequency and intensity of care required for optimal outcomes.10,12,13,16 For patients caught in this gap, those who need modified compression but who cannot afford or access regular therapy, alternative solutions are urgently needed.10,17
Extending therapeutic benefit for patients beyond the clinic requires compression options that provide effective therapeutic pressure during daytime upright positions and gentle support at night. An elastic nighttime garment (Comfiwave; Haddenham, LLC) (ENG) and an adjustable wrap device (easywrap, Haddenham, LLC) (AWD) work together to meet these needs. Although several nighttime garments incorporate LAPP-based designs, the ENG described in this analysis represents a specific low-profile, highly elastic construction that differs from bulkier nighttime options and should not be considered interchangeable with all nighttime garments. This combination supports self care with targeted compression by day, restorative therapy at night, and the flexibility to adjust fit between visits, thereby reducing reliance on frequent clinic interventions while keeping therapists involved.
The ENG evaluated for this series is a patented differential-pressure garment engineered with a LAPP and delivers a therapeutic pressure range of 15 mm Hg to 21 mm Hg for patients with primary or secondary lymphedema and chronic edema (Figure 1A). Although several nighttime garments incorporate LAPP‑based designs, as previously noted this particular ENG is distinguished by its low-profile, highly elastic construction, which differs from bulkier or nonelastic nighttime options. It may be used as a stand‑alone intervention or may be combined with additional compression when higher therapeutic pressures are indicated. Although marketed primarily for nighttime wear, it has been successfully used during both day and night, allowing flexibility for individualized care.18 Designed for extended wear, the garment can remain in situ for up to 24 hours, with removal recommended for skin care.
Constructed as a flat-knit garment, the ENG provides gentle tissue loading and micromassage through its LAPP structure without compromising comfort. This differential pressure effect has been clinically associated with improvements in integument health, including softening of fibrotic tissue and improving overall tissue texture.18 Low-resistance yarns facilitate ease of donning and doffing, while the single-layer knit structure combines elasticity with breathability.17 This latex-free garment is available in multiple configurations for the upper and lower extremities and trunk, allowing clinicians to tailor selection to individual patient needs.
The AWD evaluated in the present study is a short-stretch device constructed with overlapping, knitted bands and hook-and-loop fasteners on 1 side, rather than laminated sheet with straps (Figure 1B). Its textile bands are engineered to be conforming and low-profile, delivering adjustable, gradient compression that targets both venous and lymphatic contributors to chronic edema. The application method uses the standard 50% overlap of traditional short stretch bandages, and the specially woven knitted bands deliver consistent dosage, reducing application errors. The knitted construction provides controlled elasticity, with a tactile “lock-out” point. This feature simplifies correct application and promotes consistent graduated compression without the need for a pressure gauge. To eliminate guesswork, this device is offered in 2 therapeutic compression ranges—20 mm Hg to 30 mm Hg (light), and 30 mm Hg to 40 mm Hg (strong)—providing accurate dosing for clinical needs. In comparative testing across 7 adjustable wrap systems, the strong version demonstrated standout performance, with one of the lowest variations in lying pressure, the highest mean stiffness index, and the greatest elongation ratio, underscoring its ability to deliver both reliable pressure consistency and superior adaptability to limb shape.19
Both garments are washable and reusable. The ENG carries a manufacturer guarantee of 1 year, and the AWD is guaranteed for 6 months, reflecting typical replacement intervals for daytime and nighttime compression devices.
When the ENG is paired with the AWD during waking hours, the combination delivers a layered approach to compression therapy. The nighttime garment provides a hygienic, padded base that enhances comfort and skin integrity while stimulating the dermal lymphatic interface through its alternating pressure profile. The AWD complements this by delivering therapeutic compression dosage during upright positions, offering a dynamic, patient centered alternative to traditional compression garments. Both devices are designed for patient self application, thus reducing dependence on skilled providers and enhancing accessibility. Beyond serving as a base layer, the nighttime garment can also be used as an additional compression layer over traditional compression garments, enhancing therapeutic effect and accommodating individual patient needs. This approach has brought increased awareness to underrecognized lymphedema in wound care and chronic venous disease populations, which share overlapping pathophysiologic features and often respond to similar compression strategies.
This case series illustrates how incorporating ENG, alone or in combination with other compression strategies, can help overcome barriers to CDT, optimize clinical outcomes, and support patient adherence. It demonstrates real-world scenarios detailing diagnoses, treatment challenges, intervention protocols, and measurable results such as volume reduction, symptom relief, and adherence, along with progression to custom garments or continued use of the chosen compression approach.
Materials and Methods
This prospective case series used a convenience sample of 5 adult patients with chronic lymphedema and/or nonhealing wounds who had demonstrated minimal or no improvement with traditional standard of care approaches, such as multilayer bandaging, pneumatic compression, and wound dressings, or were unable to tolerate these treatments. Each patient was transitioned to a regimen incorporating the ENG at nighttime and either the AWD or another compression garment layered with the ENG for daytime use, while wound care continued per clinician discretion. Limb volume was measured using circumferential tape, and wound size was assessed by ruler at baseline and follow-up visits. The same clinician performed all measurements pertaining to their individual patients. This case series reflects real-world clinical practice using US Food and Drug Administration–cleared compression products rather than a formal research protocol; no experimental procedures were performed. Health Insurance Portability and Accountability Act (HIPAA) compliance was maintained, and patients consented to treatment and the use of anonymized data and images for publication.
Results
Case 1: Late stage II lymphedema and neuromuscular complications
A 72-year-old female with late stage II lymphedema involving both lower extremities presented with swelling to the mid thigh and pitting edema (Figure 2A). Her past medical history included lipedema, morbid obesity, chronic venous insufficiency (CVI), and chronic inflammatory demyelinating polyneuropathy (CIDP), the latter contributing to impaired mobility and necessitating the use of custom-molded ankle-foot orthoses for safe ambulation. At presentation her body mass index (BMI) was 35.3 (weight, 180 lb; height, 4 ft 11 in), but historically, when she was approximately 100 lb heavier prior to gastric bypass, her BMI would have been approximately 55.5. Despite this significant weight loss, she continued to experience disproportionately large, heavy legs. She ambulated with a rollator at a modified independent level and reported lifelong struggles with weight management. Previous attempts at lymphedema control included over-the-counter compression knee-high stockings (20 mm Hg-30 mm Hg), which rolled, corded, and caused skin injury, as well as intermittent pneumatic compression (IPC), which provided only temporary relief. At baseline, both lower extremities were affected up to the hips, with distal fibrosis at the foot and ankle, putty-like proximal lower leg, and a fattier consistency proximally. The patient described constant aching, heaviness, and easy bruising, rating her pain as 7 out of 10 (0 = “no pain,” and 10 = “worst pain imaginable”) utilizing a visual analogue scale (VAS).
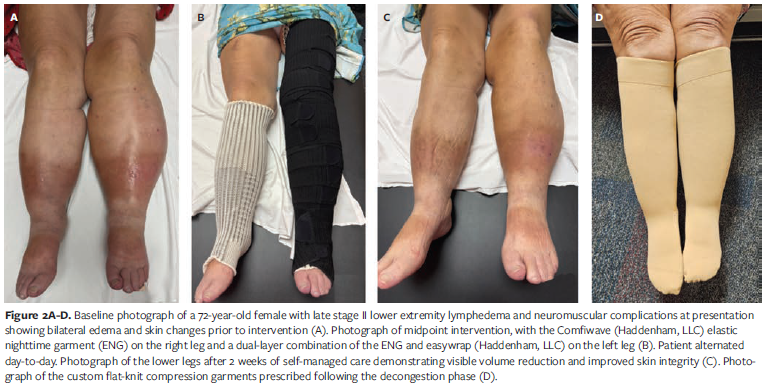
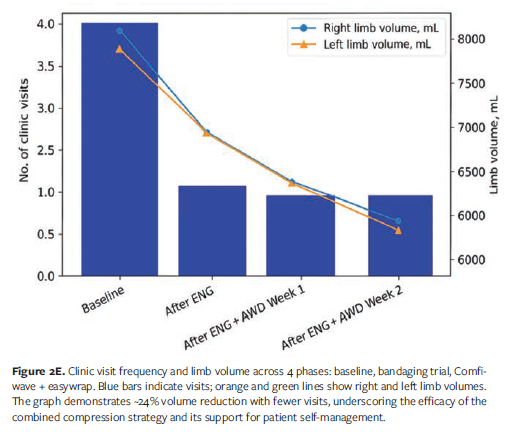
Given the patient’s underlying CIDP and associated mobility challenges, the initial approach prioritized maintaining function while assessing tolerance. Traditional multicomponent lymphedema bandaging was not an option, because the bulk of the bandage would not fit underneath her custom braces. A 2-week trial of a 2-layer, disposable, cohesive product offered some initial volume reduction; however, it had to be discontinued due to heat buildup, which exacerbated her neurogenic symptoms, and due to skin irritation caused by perspiration. The ENG was introduced on the fifth visit, and the patient returned 1 week later for reassessment. Compression was applied asymmetrically using ENGs of different lengths—1 thigh-high and 1 knee-high—worn continuously, day and night, with sides alternated daily. The garments fit comfortably under braces and shoes and did not contribute to overheating. At the next visit, the AWD was added over the ENG during upright waking, hours, and the ENG alone was used at rest at night (Figure 2B). This combined approach resulted in a marked improvement in tissue texture and limb volume (Figure 2C): The limb volume decreased from 8 056 mL (right) and 7 932 mL (left) at baseline to 6 123 mL and 6 028 mL, respectively, an approximate 24% reduction (Figure 2E). Clinic visits were reduced from 2x/wk during the bandaging phase to only 1 visit every other week during the ENG and AWD dual therapy phase, highlighting both clinical efficacy and ease of patient self management. Pain scores improved from 7/10 to 1/10 assessed with a 10 point visual analogue scale (VAS), and the patient reported, “My legs have never been this small,” noting improved mobility, transfers, and the ability to wear shoes she had not worn in years. After 2 weeks of successful self-care with this combination, the patient was measured for custom flat-knit knee-high garments for daytime use (Figure 2D). She continued to wear the ENG overnight to maintain therapeutic dermal lymphatic stimulation promoting improved integument health. In total, the patient completed 7 visits, with more than half occurring before initiating treatment with the ENG and AWD.
The ease of garment application and the patient’s ability to achieve modified independence allowed for reduced visit frequency and effective home management. Upon achieving stable volume, she transitioned to a custom-knit knee-high garment for daytime use and ENG for nighttime use. To provide variety and maintain adherence, a second ENG was ordered for nighttime compression, 1 knee-high and 1 thigh-high, giving the patient options for long-term management. She continues to alternate between flat-knit and AWD based on preference for daytime compression.
Case 2: Unilateral amputee with secondary lymphedema, CVI, and recalcitrant venous leg ulcer
A 68 year old male with advanced lower extremity lymphedema presented with a positive Stemmer sign, woody fibrotic tissue changes at the ankle, firm putty like consistency to the distal thigh, hemosiderin staining, brawny swelling with dynamic refill, and a recalcitrant venous leg ulcer measuring 3.2 cm × 2.8 cm. His past medical history included CVI and prosthesis use following prior limb trauma, contributing to gait challenges and difficulty maintaining compression. He reported discomfort, difficulty sustaining compression between visits, and challenges with prosthetic fit. Traditional multicomponent lymphedema bandaging was not feasible, because the bulk interfered with prosthetic alignment and impaired gait. A 2-week trial of 2-layer cohesive wraps offered some initial improvement but was discontinued due to bandage slippage, skin irritation, and drainage buildup, which enlarged the wound to 4.0 cm × 3.2 cm (Figure 3A). Given these barriers, the treatment plan prioritized mobility and wound care while ensuring effective compression.
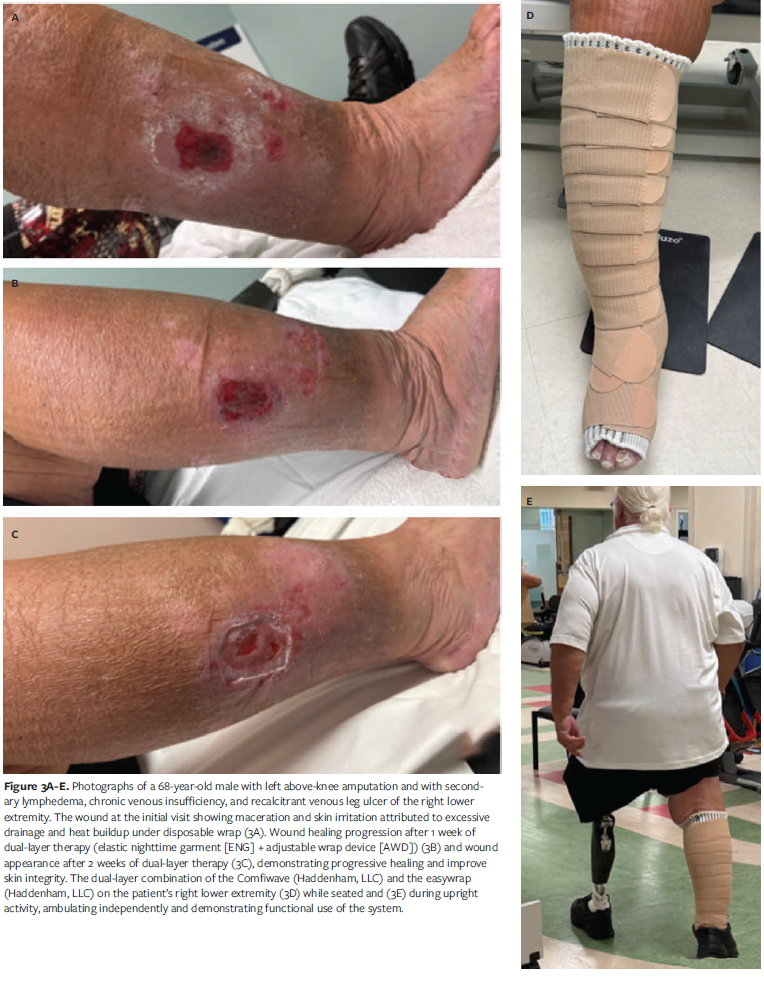
A thigh-high ENG was introduced for nighttime use, combined with a below knee ENG and the AWD during waking hours to support ambulation and accommodate prosthesis use (Figures 3D and 3E). IPC was continued over the thigh-high ENG 1 hour daily. This combination delivered stable, breathable compression without compromising gait mechanics, and the patient was able to shower and manage wound care independently between visits. After 1 week, limb volume decreased from 8 635 mL to 7 900 mL, and the wound size reduced to 3.5 cm × 3.0 cm (Figure 3B). At 2 weeks, the wound measured 2.8 cm × 2.5 cm, and by week 3, limb volume was 7 803 mL, with wound dimensions of 2.0 cm × 2.0 cm (Figure 3F). At that point, the patient was measured for a custom flat knit garment. By week 5, the wound had fully healed, and limb volume stabilized at 7 790 mL—an approximate 10% reduction from baseline (Figure 3F).
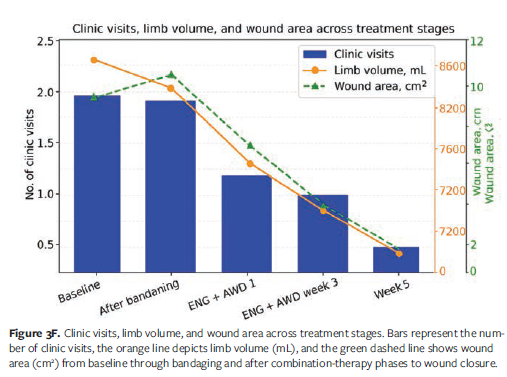
In total, the patient completed 7 visits, with more than half occurring before initiating therapy with the ENG and AWD. During the bandaging phase, he required 4 visits over 2 weeks; after transitioning to the garment combination, visit frequency dropped to weekly and then every 2 weeks until closure. Independence with garment application enabled reduced clinic visits and improved mobility and comfort. The patient reported, “I can walk better and manage my care without help.”
Case 3: Healing in the dark: Compression success in a blind patient with recalcitrant ulcers
A 71 year old male with stage III lymphedema presented with long-standing bilateral lower extremity wounds and CVI (Figure 4A). He has congenital blindness and craniocervical abnormalities, and his medical history was further complicated by severe arthritis of the left shoulder, which significantly limited his range of motion. On physical examination, tissue texture was woody from the dorsum of the foot to below the knee, with weeping anterior lower legs. Progressive swelling had begun in 2008 after decades of intermittent wound care, managed with silver antimicrobial alginate dressings under an Unna boot (zinc impregnated semirigid bandage) and cohesive bandage overlay. After the ulcers healed, traditional hook-and-loop fastener wraps with hybrid sock were prescribed; however, the patient reported no training in application or duration of use. By the time of referral for CDT, he had endured recurrent ulcerations dating back to 1990 and remained wheelchair bound for long distances due to mobility limitations. He required custom shoes because of swelling at the dorsum of the foot, which prevented use of standard footwear.
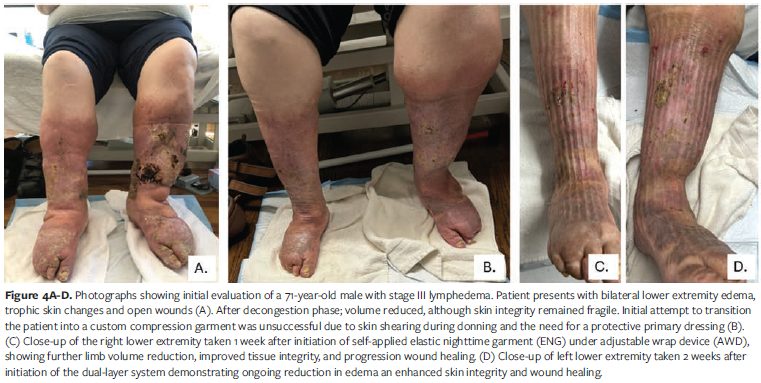
Several barriers complicated self-management. Blindness, obesity, limited mobility, reduced shoulder range of motion, and decreased upper body strength made garment application difficult. A large pannus further restricted access to the lower extremities. Custom flat-knit garments proved impractical, because they were difficult to don over wound dressings or protective layers. Traditional hook-and-loop fastener systems did not provide adequate compression at the foot and ankle while accommodating footwear necessary for safe transfers, because the hybrid sock could not fit the enlarged dorsal foot.
Given these barriers, the treatment plan prioritized wound closure, skin protection, and functional compression. An intensive CDT phase was initiated: twice weekly for 1 month, then once weekly for 4 months. A primary dressing consisting of a 2-layer hydrofiber infused with silver was applied directly over wounds and ABD pad secure with rolled guaze. A compression application consisting of an open cell foam roll, 3 short-stretch bandages, and 1 long-stretch bandage secured with the AWD. Wounds were bilateral; the right closed at 2 months, and the left closed after an additional month. Bandaging continued with foam and short-/long-stretch bandages to protect fragile skin. Setbacks included cellulitis, toe amputation on the left foot, and new diagnosis of bladder cancer. Recurrent wounds were managed with petrolatum-infused gauze and sterile abdominal combine pad beneath foam and bandages.
The patient was then transitioned to a modular compression system designed for ease of application and sustained therapeutic benefit. A below-knee ENG was paired with the AWD. To protect fragile skin and healed ulcerations, an abdominal combine pad was applied directly over vulnerable areas and held with Comprefit foam (Sigvaris; high-density, moisture-
wicking chip foam liner) beneath the ENG, with the AWD providing adjustable compression. This layered approach provided wound protection and compression stability, enabling safe, effective home-based management without reliance on frequent clinic visits.
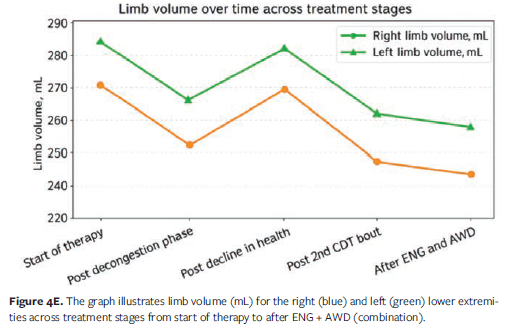
Clinical outcomes were notable (Figures 4B, C, and D). Limb volume decreased by 50% from the start of therapy, with an additional 2.78% reduction after transitioning to the ENG and AWD (Figure 4E). The compression combination allowed the patient to wear appropriate footwear, improving safety during transfers and enhancing overall mobility and confidence. Independence during the self care phase was a remarkable accomplishment given his visual impairment and physical limitations. These outcomes highlight the adaptability of garment-based compression strategies in overcoming barriers to traditional bandaging and supporting patient autonomy in complex clinical scenarios.
Case 4: Optimizing surgical outcomes: Compression therapy venous ablation
A 75-year-old female presented with stage IIb lymphedema of the right lower extremity with hyperpigmentation, positive Stemmer sign, and Bjork bow tie test findings of 75% lower leg with woody skin texture and 25% upper leg with puddy consistency (Figure 5A). She described insidious onset approximately 10 years earlier. Initial evaluation by a vascular specialist suggested lymphatic involvement rather than venous disease; however, after swelling persisted she sought a second opinion. The second provider performed 3 venous ablation procedures, after which swelling worsened in the right leg and the patient experienced ongoing difficulty accessing appropriate care. Despite being highly active and walking several miles daily, she did not achieve adequate compression with a custom flat-knit stocking (compression class 3, 34 mm Hg–46 mm Hg). Persistent refill and volume fluctuation were noted, indicating insufficient therapeutic effect from the prescribed garment.
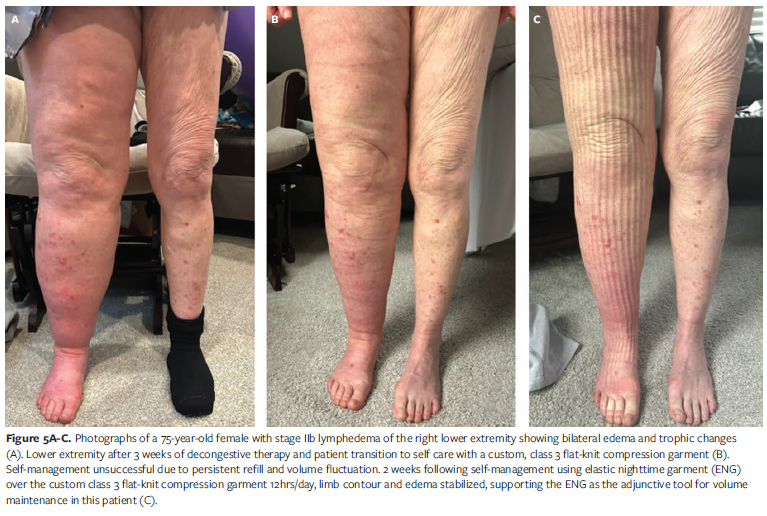
Given these barriers, the treatment plan prioritized wound closure, skin protection, and functional compression. An intensive phase of CDT was initiated using multilayer bandaging with toe wraps, open cell foam, 6 short-stretch bandages, and 2 cotton elastic long-stretch bandages secured with hook-and-loop fastener. Sessions were conducted twice weekly for 8 weeks. After 6 weeks, limb volume reduction plateaued, but swelling recurred upon transitioning to a custom flat-knit garment combined with a nighttime chopped foam garment with fluid-directing channels (Figure 5B). To regain control prior to travel, the patient incorporated a daytime combination of the ENG and AWD for 3 weeks. This approach allowed her to maintain her exercise routine, reduce clinic visits from 2 to 3 times per week to once weekly, and achieve greater independence during the intensive phase (Figure 5C). Upon re-achieving volume reduction, the patient transitioned back to the custom flat-knit garment, supplemented with a thigh-high ENG overlay for enhanced compression.
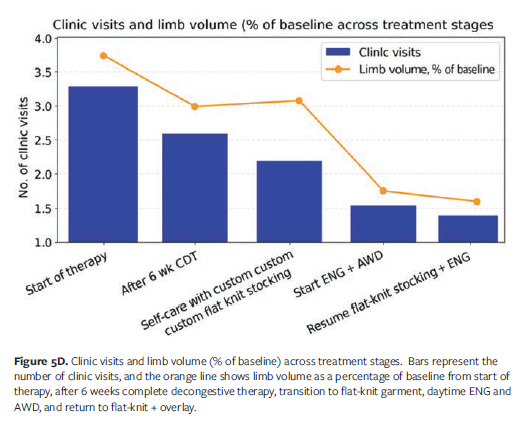
The patient experienced an additional limb volume reduction of 7.8% from the start of therapy (Figure 5D). She reported increased satisfaction with care and greater confidence in her ability to manage lymphedema independently. This case highlights the utility of adaptive compression strategies in optimizing outcomes for patients with prior surgical interventions and persistent edema, while supporting autonomy and long-term self management.
Case 5: Optimizing surgical outcomes: Compression therapy after LVA and liposuction for lymphedema
A 34-year-old female diagnosed with primary lymphedema of the right lower extremity presented with a medical history complicated by a blood clotting disorder, anxiety, and celiac disease. Approximately 5 years earlier she had undergone multiple lymphaticovenous anastomosis procedures followed by CDT, but she was advised to remove bandages between sessions, which led to recurrent swelling and dissatisfaction with follow-up care. Seeking a more effective solution, she consulted a new surgeon who recommended right lower extremity lipectomy following an intensive phase of CDT at the facility of author S.D. The patient attended therapy once weekly for 1 month prior to surgery, remaining in compression garments around the clock aside from showering, and was educated on self bandaging with the ability to remove and reapply independently. In the summer of 2025, she underwent lipectomy, followed by physical therapy twice weekly for 1 month. She was placed in a compression class 3 (34 mm Hg–46 mm Hg) flat-knit garment in the operating room but developed a wound at the anterior right ankle, prompting transition to short-stretch bandage wrapping with open cell foam. Therapy was then reduced to once weekly when the ENG and AWD were introduced in place of traditional bandaging.
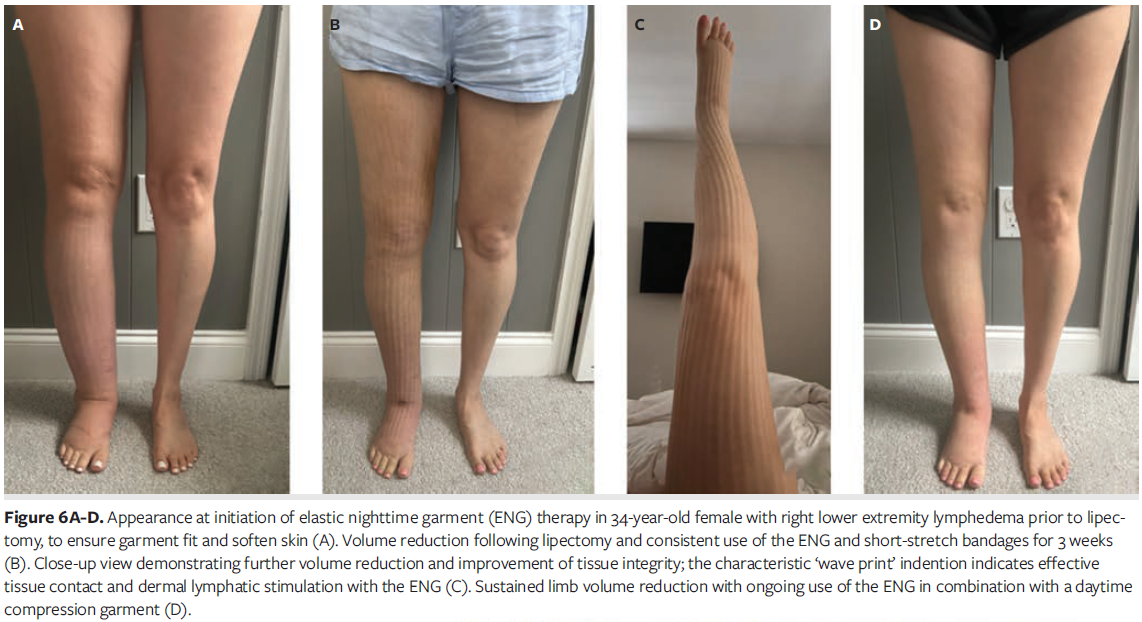
Postoperative bruising and significant pain made it difficult for the patient to don, doff, and tolerate the custom flat-knit garment as prescribed. She struggled to maintain consistent compression, particularly during sleep and daily activities, which posed a risk for rebound edema and delayed recovery. To address these challenges, she transitioned to a modular compression approach combining the ENG and AWD (Figures 6A-D). This regimen alternated between the ENG either worn alone or layered beneath 3 short-stretch bandages extending from foot to groin. The AWD enabled independent application and adjustment of compression, reducing reliance on skilled assistance. Skilled therapy sessions supplemented this strategy by providing targeted bandaging for areas of fibrosis and ensuring proper hygiene and garment care.
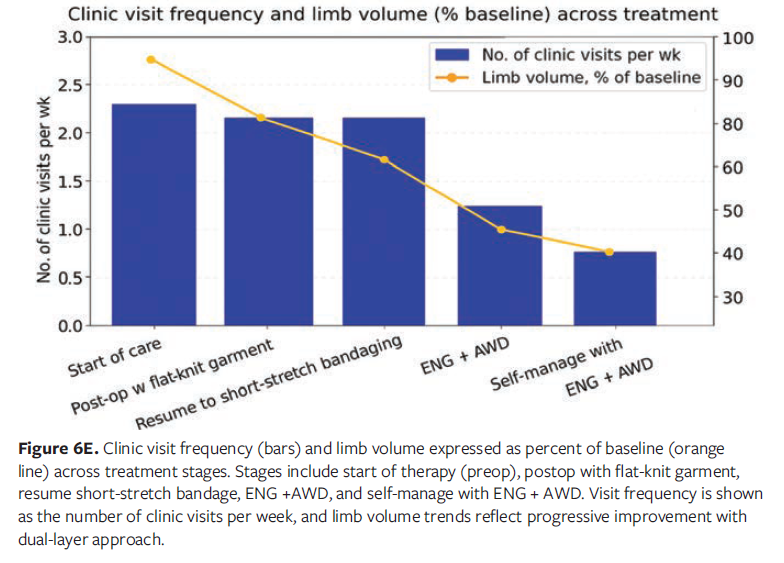
The patient achieved 15.8% limb volume reduction from the start of therapy following surgery, with an additional 4% reduction after transitioning to the ENG and AWD combination (Figure 6E). She regained safe ambulation with appropriate footwear worn over the ENG and was able to resume driving once she was medically cleared. These outcomes demonstrate the effectiveness of adaptive compression strategies in supporting postoperative recovery and promoting patient independence.
Discussion
The cases presented highlight the clinical value of combining an ENG and an AWD as an alternative or adjunctive compressive tool to traditional multilayer bandaging for patients with complex lymphedema and chronic wounds. This approach is particularly relevant for individuals facing barriers to skilled care; those for whom conventional protocols are impractical due to bulk, heat intolerance, or the need for frequent clinic visits; and those who have not achieved adequate outcomes with conservative or mainstream compression options. Rather than viewing compression as a fixed pressure target, this combination reflects a therapeutic principle, with compression as a dynamic tool responsive to tissue characteristics, functional needs, and patient lifestyle. Across diverse scenarios, including prosthetic fit challenges, visual impairment, prior venous interventions, and postoperative garment intolerance, adaptive compression strategies consistently offered by the combination of the ENG and AWD enabled independence, reduced visit frequency, and sustained volume reduction.
Nighttime compression has emerged as a critical adjunct in lymphedema management, providing sustained therapeutic pressure during rest to prevent rebound edema, improve tissue oxygenation, and support wound healing.22-24,26-28 Clinical studies of nighttime garments demonstrate measurable benefits in venous return, patient comfort, sleep quality, and long-term edema control, underscoring their importance in comprehensive care models.21-28 As evidenced in the case studies presented here, as well as in other published evaluations of nighttime compression systems, patients experience both functional and quality of life improvements when nighttime therapy is integrated into care. However, many existing nighttime systems are bulky, heat retentive, or difficult for patients to tolerate, limiting adherence despite proven efficacy.
In this context, it is important to distinguish the ENG highlighted in this case series from other low-profile textiles that also have a LAPP design, such as EdemaWear (Compression Dynamics, LLC). The open‑weave, longitudinally elastic fuzzy wale design of this product creates a microchannel effect that can assist with mild edema and is valued for comfort and ease of donning. However, its compression profile is generally lower than the therapeutic range delivered by the ENG featured in the present series (10 mm Hg-15 mm Hg vs 15 mm Hg-21 mm Hg, respectively). While both textile designs produce a LAPP capable of supporting dermal lymphatic stimulation and tissue softening, the higher and more consistent compression dose of the ENG maintains limb volume more reliably in moderate to advanced lymphedema. The cases in this series demonstrate that patients with chronic wounds, fibrosis, or higher‑stage disease often require the more structured and reliable compression provided by the ENG rather than the lighter, adjunctive support offered by the aforementioned comparison low-profile textile.
The ENG highlighted in this case series represents a significant innovation in this space, offering a lower-profile textile solution constructed from a patented knitted fabric with elastic yarn and breathable cotton. Its uneven mesh creates microchannels that promote lymphatic flow and tissue softening, delivering gentle, sustained compression suitable for 24-hour wear. Unlike traditional nighttime garments that are restricted to use during rest, this specific ENG can also be worn comfortably during the day, stabilizing limb volume between therapy sessions and reducing the risk of rebound swelling. This dual functionality makes it a standout option, combining the evidence-based benefits of nighttime compression with the practicality of daytime use, thereby expanding patient autonomy and improving adherence across diverse clinical scenarios.
The AWD complements the ENG by providing adjustable short-stretch mechanics that deliver low resting and high working pressures, optimizing comfort at rest and therapeutic effect during movement. The engineered lockout system of the AWD ensures consistent graduated compression without requiring precise strap tension, thus reducing variability and improving predictability in clinical outcomes. Although the particular AWD studied in the present series is classified within the adjustable hook-and-loop fastener wrap category, it is distinguished by its knitted textile construction rather than the traditional laminated strap design. This knitted design provides greater elasticity, breathability, conformability, and consistency of compression, setting it apart from conventional wraps and enhancing both comfort and therapeutic reliability.19 These distinctions are clinically meaningful given the wide range of AWDs available on the market. Comparative studies further demonstrate that performance differences among adjustable wraps directly influence therapeutic dosing and venous return.19,29 In a comparative review of 7 hook-and-loop fastener adjustable wraps, the proprietary strong version mentioned in the introduction of this article showed superior biomechanical properties, including the second lowest variation in lying pressure at 5.9 mm Hg, the highest mean static stiffness index at 13.2 mm Hg, and the greatest mean elongation ratio at 137%, outperforming overall standard deviation of 8.9 mm Hg and overall mean 9.5 mm Hg and 107.1%, respectively.19 These metrics matter because consistent pressure ensures predictable outcomes, increased stiffness enhances venous function and edema reduction, and greater tensile strength maintains compression longer without frequent readjustment.31,32 In contrast, wraps with higher stretch allow greater resting pressures but lower stiffness, thus reducing dynamic efficacy.31
Clinical evidence further supports an AWD, with case studies reporting rapid wound epithelialization within 1 month and complete closure within 6 weeks when the AWD highlighted in this case series was used to replace bandaging, alongside measurable limb volume reductions.32 In this case series published in 2019, the AWD used in current case series was shown to offset its initial cost within 1 month when used instead of bandaging 3 times per week, and by 6 weeks when compared with a 2-layer short-stretch system.33
These outcomes reflect improved patient autonomy, daily hygiene, and concordance—factors essential for chronic wound healing and sustainable lymphedema management. Taken together, the comparative performance data and case-based outcomes illustrate how adaptive compression solutions not only deliver measurable clinical and economic benefits but also establish a foundation for resilient care models.19,32,33
This resilience became particularly evident during the COVID-19 pandemic, when traditional decongestive lymphedema treatment protocols were disrupted and patient-led strategies proved essential. Adjustable wraps such as the AWD studied in the present series enabled continuity of care under virtual supervision, reducing clinic visits and maintaining limb volume control.34 These innovations validated the role of adaptive compression in decentralized care models and reinforced the need for solutions that combine clinical efficacy with patient autonomy. Together, these findings highlight a shift from compression as a fixed pressure target to compression as a dynamic, patient-centered tool. By combining textile innovation with adjustable mechanics, the ENG and AWD studied in the present series expand access, reduce resource burden, and empower patients to sustain outcomes in real-world settings. Further research is warranted to standardize performance metrics, refine selection criteria, and explore long-term outcomes across diverse patient populations.
Limitations
This case series has several limitations. First, the small sample size of 5 patients restricts the generalizability of the findings and may not reflect outcomes in the broader population of individuals with lymphedema or chronic wounds. Second, the case-based design is descriptive and observational rather than controlled or randomized; without a comparison group, improvements cannot be attributed solely to the intervention. Reported gains in comfort, independence, and satisfaction are based on patient feedback rather than on validated quality-of-life instruments, which limits interpretability. Additionally, the cases included varied diagnoses, comorbidities, and functional limitations. While this reflects real-world complexity, it introduces variability that may influence outcomes. Most cases report results over several weeks rather than months or years; thus, long-term durability of limb volume reduction and adherence to garment use is unknown. Concurrent interventions such as wound care and skilled therapy may also have contributed to improvements, making it difficult to isolate the effect of the garment combination.
Finally, limb volume was measured using circumferential tape, which is subject to operator variability. Although the same clinician performed all measurements to enhance intrarater reliability, more precise methods such as perometry or bioimpedance could have provided greater accuracy.
Conclusion
This case series demonstrates the clinical utility of a modular, textile-based compression strategy for patients with complex lymphedema and chronic wounds who face barriers to traditional multilayer bandaging or single compression garments. Taken together, the comparative performance data and case-based outcomes illustrate how adaptive compression solutions, specifically the unique knitted band design of the AWD combined with the lower-profile textile innovation of the ENG, deliver measurable clinical and economic benefits, while establishing a foundation for resilient care models that can withstand disruptions in traditional service delivery.
As compression therapy continues to evolve, adaptable systems that prioritize comfort, independence, and clinical efficacy may play a critical role in improving adherence and reducing treatment burden, particularly in underserved or resource-limited settings. Although these findings are promising, further research with larger cohorts, longer follow-up periods, and standardized outcome measures is warranted to validate efficacy, assess long-term durability, and inform best practice guidelines.
Author and Public Information
Authors: Suzie Ehmann, PhD, DPT, CWS, CLT-LANA1; and Sandi Davis, PT, DPT, CLT-LANA, CLWT2
Affiliations: 1McLeod Regional Medical Center - Seacoast, Little River, SC, USA; 2Davis Care Physical Therapy New York, NY, USA
Disclosure: The authors declare a financial relationship with one or more companies that produce or market products or services relevant to the topic of this manuscript. Specifically, S.E. discloses the following financial relationship(s): Clinical Specialist – Haddenham, LLC; Paid Speaker, advisory board member and clinical consultant: Compression Dynamics, LLC; Urgo Medical North America; L&R, USA, Inc; Ovik Health; and Medline, Inc. S.D. is a paid consultant for Koya Medical, Inc.
Correspondence: Sandi Davis, PT, DPT, CLT-LANA, CLWT. Davis Care Physical Therapy, 8 West 36th Street, 6th floor, New York, NY 10018; PT@DavisCarePT.com
Manuscript Accepted: January 14, 2026
References
- Executive Committee of the International Society of Lymphology. The diagnosis and treatment of peripheral lymphedema: 2020 consensus document of the International Society of Lymphology. Lymphology. 2020;531:3-19. doi:10.2458/lymph.4649
- Campione E, Jackson K, Hunley JH. Lymphoedema practice patterns: maintenance recommendations. Journal of Lymphoedema. 2021;161:33-38.
- Executive Committee. The diagnosis and treatment of peripheral lymphedema: 2016 consensus document of the International Society of Lymphology. Lymphology. 2016;494:170-184.
- Zasadzka E, Trzmiel T, Kleczewska M, Pawlaczyk M. Comparison of the effectiveness of complex decongestive therapy and compression bandaging as a method of treatment of lymphedema in the elderly. Clin Interv Aging. 2018;13:929-934. doi:10.2147/CIA.S159380
- Bergmann A, Baiocchi JMT, de Andrade MFC. Conservative treatment of lymphedema: the state of the art. J Vasc Bras. 2021;20:e20200091. doi:10.1590/1677-5449.200091
- Bjork R, Ehmann S. S.T.R.I.D.E. professional guide to compression garment selection for the lower extremity. J Wound Care. 2019;28(Suppl 6a):S1-S44. doi:10.12968/jowc.2019.28.Sup6a.S1
- Ehmann S, Walker KJ, Bailey CM, DesJardins JD. Experimental simulation study to assess pressure distribution of different compression applications applied over an innovative primary wound dressing. Wounds. 2020;3212:353-363.
- Ashforth K, Morgner S, VanHoose L. A new treatment for soft tissue fibrosis in the breast. Journal of Lymphoedema. 2011;62:42-46.
- Moffatt CJ, Doherty DC, Franks PJ, Mortimer PS. Community-based treatment for chronic edema: an effective service model. Lymphat Res Biol. 2018;161:92-99. doi:10.1089/lrb.2017.0021
- Ehmann S, McKeown B, Davis S, Bock KJ, Whitaker JC, Dolgoy N. Using the STRIDE algorithm for compression selection in upper-body lymphoedema. J Wound Care. 2025;34(Suppl 11c):S36-S48. doi:10.12968/jowc.2025.34.S11c.S36
- Ehmann S. The Biophysical Impact of an Alternating Compression Profile. Dissertation. Nova Southeastern University; 2024.
- Bian J, Shen A, Yang W, Zhang L, Qiang W. Financial toxicity experienced by patients with breast cancer-related lymphedema: a systematic review. Support Care Cancer. 2023;316:354. doi:10.1007/s00520-023-07800-9
- Mahmoudi M, Gould LJ. Opportunities and challenges of the management of chronic wounds: a multidisciplinary viewpoint. Chronic Wound Care Management and Research. 2020;7:27-36. doi:10.2147/CWCMR.S260136
- Executive Committee of the International Society of Lymphology. The diagnosis and treatment of peripheral lymphedema: 2023 consensus document of the International Society of Lymphology. Lymphology. 2023;564:133-151.
- Centers for Medicare & Medicaid Services. Lymphedema compression treatment items. Accessed November 30, 2025. https://www.cms.gov/medicare/payment/fee-schedules/durable-medical-equipment-prosthetic-devices-prosthetics-orthotics-supplies/lymphedema-compression-treatment-items
- Keast DH, Despatis M, Allen JO, Brassard A. Chronic oedema/lymphoedema: under-recognised and under-treated. Int Wound J. 2015;123:328-333. doi:10.1111/iwj.12224
- Phillips N, Lawrance S. Haddenham Comfiwave: a unique compression device for lymphoedema treatment. Br J Community Nurs. 2020;25(Suppl 4):S23-S30. doi:10.12968/bjcn.2020.25.Sup4.S23
- Ehmann S, McKeown B, Davis S, Bock KJ. The science of compression textiles and garments for upper-body lymphoedema. J Wound Care. 2025;34(Suppl 11c):S19-S29. doi:10.12968/jowc.2025.34.S11c.S19
- Phillips N, Wright T. Comparing the variation of pressure, stiffness and elasticity achieved in seven adjustable wrap systems including Easywrap Strong. J Wound Care. 2024;33(Suppl 1):S10-S14. doi:10.12968/jowc.2024.33.Sup1a.S10
- Ramadan F. Night-time compression: a valuable addition to the lymphoedema management toolkit. Br J Community Nurs. 2023;286:293-296. doi:10.12968/bjcn.2023.28.6.293
- McNeely ML, Dolgoy ND, Rafn BS, et al. Nighttime compression supports improved self-management of breast cancer–related lymphedema: a multicenter randomized controlled trial. Cancer. 2022;1283:587-596. doi:10.1002/cncr.33943
- Mazur S, Szczęśniak D, Tchórzewska-Korba H. Effectiveness of Mobiderm Autofit in the intensive phase of breast cancer-related lymphedema treatment: a case series. Lymphat Res Biol. 2023;216:608-613. doi:10.1089/lrb.2022.0079
- Brunelle CL, Taghian AG. The important role of nighttime compression in breast cancer-related lymphedema treatment. Cancer. 2022;1283:458-460. doi:10.1002/cncr.33942
- Bertsch T. Evaluation of a novel night-time compression garment: a prospective observational study. Br J Community Nurs. 2018;2311:535-541. doi:10.12968/bjcn.2018.23.11.535
- Mosti G, Cavezzi A. Compression therapy in lymphedema: between past and recent scientific data. Phlebology. 2019;348:515-522. doi:10.1177/0268355518824524
- Bock KJ, Muldoon J. A 24-hour interval compression plan for managing chronic oedema: part 1—the science and theory behind the concept. J Wound Care. 2022;31(Suppl 2):S4-S9. doi:10.12968/jowc.2022.31.Sup2.S4
- Mestre S, Calais C, Gaillard G, et al. Interest of an auto-adjustable nighttime compression sleeve (Mobiderm Autofit) in maintenance phase of upper limb lymphedema: the MARILYN pilot RCT. Support Care Cancer. 2017;258:2455-2462. doi:10.1007/s00520-017-3652-5
- Whitaker JC. Lymphoedema management at night: views from patients across five countries. Br J Community Nurs. 2016;21(Suppl 10):S22-S30. doi:10.12968/bjcn.2016.21.Sup10.S22
- Benigni JP, Uhl JF, Filori P, Balet F, Penoel L. Adjustable compression wraps: stretch, interface pressures and static stiffness indices. Int Angiol. 2023;423:247-253. doi:10.23736/S0392-9590.23.04957-X
- Mosti G, Partsch H. A new two-component compression system turning an elastic bandage into an inelastic compression device: interface pressure, stiffness, and haemodynamic effectiveness. Eur J Vasc Endovasc Surg. 2018;551:126-131. doi:10.1016/j.ejvs.2017.09.019
- Mosti G, Mattaliano V, Partsch H. Inelastic compression increases venous ejection fraction more than elastic bandages in patients with superficial venous reflux. Phlebology. 2008;236:287-294. doi:10.1258/phleb.2008.008009
- Lee N, Lawrance S. Haddenham Easywrap: an alternative to compression bandaging in chronic oedema and wound care. Br J Community Nurs. 2019;24(Suppl 4):S22-S28. doi:10.12968/bjcn.2019.24.Sup4.S22
- Lee N, Pugh S, Cooper R. Haddenham Easywrap as part of self-management in lymphoedema and lipoedema: the patient perspective. Br J Community Nurs. 2017;22(Suppl 10):S50-S57. doi:10.12968/bjcn.2017.22.Sup10.S50
- Everett J, Lawrance S, Phillips N. Rethinking decongestive lymphoedema treatment during the pandemic. Br J Community Nurs. 2021;26(Suppl 4):S16-S22. doi:10.12968/bjcn.2021.26.Sup4.S16
















